Devices
Pacemakers
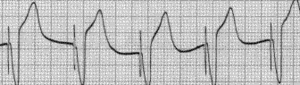
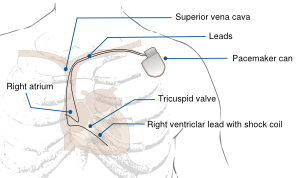
A pacemaker monitors the electrical impulses in the heart. When needed, it sends small electrical impulses to the heart muscle to maintain a normal heart rate.
A pacemaker rhythm can easily be recognized on the ECG. It shows pacemaker spikes: vertical signals that represent the electrical activity of the pacemaker. Usually these spikes are more visible in unipolar than in bipolar pacing (see image on the right).
Patients
In general, pacemaker implantation is indicated in patients with a documented irreversible bradycardia which causes symptoms. Symptoms associated with bradycardia can be lightheadedness, palpitations, dyspnea, angina and syncope. The causal relation between the abnormal heart rhythm and symptoms is crucial for the decision to implant a pacemaker.
Implantation
Pacemaker implantation is performed under local anaesthesia in a sterile operating room or catheterization laboratory. Implantation starts with inserting the pacemaker lead(s), most often into the left or right cephalic vein. Fluoroscopy is used to ensure the appropriate location in the atrium or ventricle. Leads are often positioned in the auricle of the right atrium and/or the right ventricular apex.
Device measurements
| Ohm’s law: |
|---|
| U = I x R
I = Current R = Impedance |
Stimulation threshold (in mV@ms):
The stimulation threshold is the minimum stimulus intensity (in mV) and duration (in ms) necessary to capture the heart.
Sensitivity (in mV):
The degree that the pacing system “sees” or senses signals, controlled by the sensitivity setting which is graduated in mV.
Impedance (in Ω):
The total pacing impedance is the sum of all resistance to the flow of the electrical impulses.
Battery:
Device longevity is estimated based on the remaining battery voltage and battery impedance (and past history of percent pacing in case battery status is checked at follow up visits).
If these tests are within limits, the pacemaker leads are properly positioned. Thereafter, the pacemaker pulse generator is implanted subcutaneously or subpectorally under the left or right clavicle.
Programming
Pacemaker codes
Pacemakers can be categorized according to the NASPE coding system that usually consists of 3-5 letters.
| I | II | III | IV | V |
|---|---|---|---|---|
| Chamber(s) paced
O = none A = atrium V = ventricle D = dual (A+V) |
Chamber(s) sensed
O = none A = atrium V = ventricle D = dual (A+V) |
Response to sensing
O = none T = triggered I = inhibited D = dual (I+T) |
Rate modulation
O = none R = rate adaptive |
Multisite pacing
O = none A = atrium V = ventricle D = dual (A+V) |
Explanations of table:
III:
Triggered: |
A sensed event triggers a pacemaker output pulse
Inhibited: |
Detection of physiological heart activitity will inhibit an electrical pacemaker impulse
Dual: |
A pacemaker with dual response to sensing will inhibit a pacemaker output pulse if it senses an intrinsic event in that same chamber, but it will trigger a pacemaker output pulse in the ventricle if it senses an intrinsic event in the atrium (after a programmed atrioventricular interval)
IV:
Rate modulation: |
In some patients, rate adaptive pacing is programmed “on” to ensure that when patients exercise increases, the pacemaker ensures that the heart rate increases to provide additional cardiac output. There are many ways to sense physiological exercise, including motion sensors and ventilation sensors.
Commonly used pacemakers
AAI:
The atria are paced, when the intrinsic atrial rhythm falls below the pacemaker's threshold.
VVI:
The ventricles are paced, when the intrinsic ventricular rhythm falls below the pacemaker's threshold.
VDD:
The pacemaker senses atrial and ventricular events, but can only pace the ventricle. This type of pacemaker is used in patients with a reliable sinus node, but with an AV-block.
DDD:
The pacemaker records both atrial and ventricular rates and can pace either chamber when needed.
Uni- and bipolar sensing and stimulation
Sensing and stimulation of the myocardium demands a closed electrical circuit. A pacemaker can sense and stimulate in a unipolar and bipolar fashion:
Unipolar:
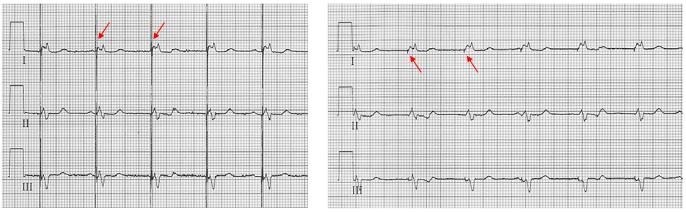
Pacemaker circuit between 1 electrode at the distal end of lead and the pacemaker pulse generator (large circuit ± 40-60 cm).
- Advantage: large pacemaker spikes (easier interpretation pacemaker ECG)
- Disadvantage: extracardiac stimulation (pectoral muscle), sensing of extracardiac signals (such as ventricular depolarisations in the atrial sensing channel (far field R waves) or non-physiological noise)
Bipolar:
Pacemaker circuit between 2 electrodes at the distal end of the lead (small circuit ± 10-15 mm)
- Advantage: more reliable sensing
- Disadvantage: small pacemaker spikes (difficult interpretation pacemaker ECG)
Follow up
The first 6-8 weeks after implantation, patients are advised not to over-stretch their arm on the same side as the pacemaker (such as golf, swimming etc.) to allow time for the lead to mature.
Control visits for pacemakers are usually every 6 months. During this visit several electrical parameters are measured: battery status, stimulation thresholds and impedance.
Complications
Undersensing:
An intrinsic depolarization that is present, is not sensed by the pacemaker. This can be due to:
- Inappropriately programmed sensitivity
- Lead dislodgement
- Lead failure, such as lead fracture or insulation failure
- Lead maturation: the amplitude may abruptly decline during the first week after implantion, but these values return to the implantation values after about 6-8 weeks as the lead “matures”
Oversensing:
The pacemakers senses signals on the marker channel that do not correspond to the ECG pattern. This can be physiologic (e.g. ventricular pulse or myopotentials) or non-physiologic (e.g. lead fracture or if the lead is loose from the pacemaker pulse generator or outside interference such as TENS therapy or surgical diathermy).
Non-capture:
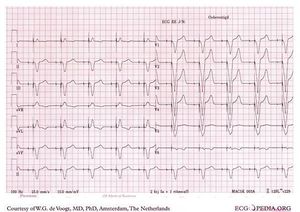
The electrical impulse is not followed by the myocardium.
This can be due to:
- Lead dislodgement
- Cardiac perforation
- Poor connection between lead and pacemaker
- Lead maturation: as the lead matures and becomes surrounded by fibrotic tissue, the threshold of stimulation decreases, which may result in non-capture
- Twiddler’s syndrome: a permanent malfunction of a pacemaker due to the patient's manipulation of the pulse generator
- Electrolyte disturbances: hyperkalemia, acidosis and alkalosis can affect the stimulation threshold
- Myocardial infarction: if a MI occurs near the tip of the head, an increase in stimulation threshold and/or non-capture can occur
- Drug therapy: e.g. flecainide can affect the stimulation threshold
- Battery depletion: if the delivered voltage is significantly reduced, advanced stages of battery depletion may result in non-capture
- Exit block: occurs when the stimulation threshold exceeds the pacemaker’s maximum output
Changes in impedance:
High lead impedance can be due to an open pacing circuit (e.g. lead is not connected to the pacemaker pulse generator or lead fracture). Low lead impedance can be due to a insulation break, which exposes the wire to body fluids which have a low resistance. Also, fluid/blood in the pacemaker header can decrease the lead impedance.
Pacemaker syndrome:
Pacemaker syndrome is the presence atrioventricular (AV) dissynchrony occurs after pacemaker implantation, regardless of the pacing mode. It is an iatrogenic disease that causes symptoms of fatigue, lightheadedness and hypotension. Symptoms are caused by the loss of atrial contribution to ventricular filling leading to a combination of backward and forward failure. Also atrial cannon waves can be present. In most cases treatment consists of adding an atrial lead and optimizing AV synchrony.
Operative failures:
Implantation related complications are:
- Pneumo/hematothorax (may require drain)
- Cardiac perforation/tamponade (may require drain)
- Pericarditis
- Lead dislodgement
- Infection: pocket infection or lead- or pacemaker pulse generator infection (may require pacemaker extraction)
- Bleeding/hematomas (may require drainage)
Long term complications:
Long term pacemaker related complications are:
- Erosion of the pulse generator through the skin (requires pacemaker extraction)
- Venous thrombosis (vena cava superior syndrome, deep vein thrombosis, lung embolism)
Implantable cardioverter defibrillators (ICD)
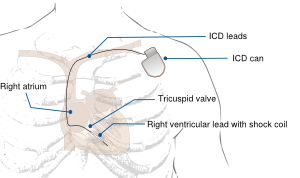
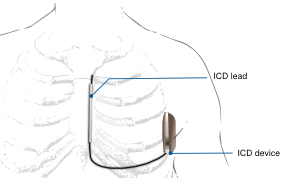
An ICD is a device that monitors heart rhythms. If it senses dangerous rhythms, it delivers shocks or anti-tachypacing (ATP) therapy. Many ICDs record the heart's electrical patterns when there is an abnormal heartbeat.
Nowadays, transvenous ICDs can also deliver pacing therapy if necessary. If a patient does not have an indication for pace-therapy, the pacemaker in the ICD is programmed at VVI 40 bpm (see above pacemaker programming).
Patients
Many ICD trials have been performed to establish the ICD indications, particularly in ischemic and dilated cardiomyopathy. Indications for ICD therapy can be divided into secondary and primary prevention. Basically, the indications are expanding from secondary to primary prevention, depending on the underlying heart disease.
Secondary prevention
Secondary prevention is therapy for patients who have already suffered a cardiac arrest or syncopal/hypotensive ventricular tachycardia. All patients who are resuscitated from VT/VF or who experienced spontaneous hemodynamic non-tolerated sustained VT in the absence of a reversible cause (e.g. acute myocardial infarction or electrolyte disturbances) have an indication for ICD therapy, regardless of the type of underlying heart disease.
Primary prevention
Primary prevention is therapy that is given in order to prevent sudden death in patients who have not yet suffered a life-threatening sustained ventricular arrhythmia, but who are at high risk of such an arrhythmia. ICD therapy is effective in patients with a low left ventricular ejection fraction (LVEF) =30% more than one month after myocardial infarction that never had shown any ventricular arrhythmia (MADITT II trial). Additionally ICD therapy appeared to be effective in patients with ischemic or dilated cardiomyopathy and a LVEF of =35% and congestive heart failure NYHA class II or III.
Patients do not meet the evidence based ICD implantation criteria if they have (1) a myocardial infarction within 40 days before ICD implantation; (2) newly diagnosed heart failure at the time of ICD implantation without prior therapy; (3) NYHA class IV symptoms of congestive heart failure.
A seperate chapter deals with a more complete list of ICD indications
Implantation
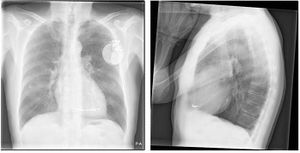
ICDs are implanted under local anaesthesia in a sterile operating room or catheterization laboratory. Transvenous ICD implantation starts, like pacemaker implantation, with inserting the ICD leads in the left or right cephalic vein. Fluoroscopy is used to ensure the appropriate location in the atrium or ventricle. Leads are often positioned in the auricle of the right atrium and/or the right ventricular apex.
After implantation, defibrillation testing is done by inducing VT/VF to test the ICD system and determine the defibrillation threshold.
Device measurements
Also device measurements will be done during and after ICD implantation. These are indicators of device and lead functioning and are comparable to the measurements in pacemaker systems (see above). Heart signal, sensing and pacing thresholds and lead impedance will be measured implantation:
Heart signal:
Real time intracardial electrocardiograms are measured. During follow up, sudden and significant decrease or disappearance of amplitudes and/or slew rates for P and/or R waves can be a sign of lead- or device problems and are further investigated.
Sensing and pacing thresholds:
During ICD implantation baseline sensing and pacing thresholds are measured. During follow-up the sensing and pacing thresholds will be compared to the chronic baseline. Significant increases or decreases may be indicative for lead- or device problems and are further investigated.
Lead impedance:
During ICD implantation baseline lead impedance is measured. During follow-up lead impedance is compared to the chronic baseline. Decreases of pacing impedances may be indicative of insulation failure. Sudden and significant increases in pacing impedance may be indicative of conduction fracture.
Battery status:
Battery status is determined during implantation and every follow up visit.
Programming
In order for and ICD to deliver therapy for specific tachyarrhythmias, it needs a reliable method to sort out arrhythmias, group them by categories (e.g. supraventricular tachycardia (SVT) and ventricular tachycardia (VT)) and make a determination as to when therapy delivery is mandated. Different ICD variables can be programmed in different ICDs, depending on the ICD manufacturer. Mainly, these ICD variables can be programmed:
Arrhythmia detection zones
The ICD diagnoses rhythm disorders by counting intervals on the intracardiac electrogram. This is a rate-based detection scheme that can be adjusted to meet the individual patient’s needs by programming. The ICD counts the current interval as one value and then average of the current interval and the preceding intervals. If these intervals fall into the same category, the event is binned in that category. If both events are tachycardia of fibrillation, but not in the same category, the interval is binned in the higher category.
| SVT discriminators: |
|---|
|
The arrhythmia detection zone is the category in which a predefined therapy will be given:
- Monitor zone (e.g. 160-180 bpm): All events in this zone will be recorded in the ICD and can be seen during follow up visits, however no therapy is given.
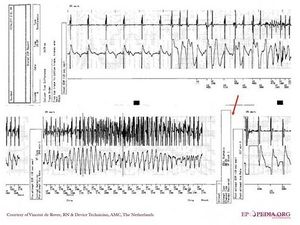
- Fast VT zone (e.g. 180-240 bpm): All events in this zone will be recorded in the ICD and can be seen during follow up visits. Therapy is given or not if the arrhythmia satisfies several criteria, which are programmed as well, such as SVT/VT discriminators.
- VF zone (e.g. >240 bpm): All events in this zone will be recorded in the ICD and can be seen during follow up visits and therapy is given immediately.
Numbers of intervals to detect / Time to detect
The ICD diagnoses an arrhythmia when a sufficient (and programmable) number of events in an event sequence are binned. This is usually stated in an “X out of Y” pattern, such as 12 out of 16 intervals.
Therapy
- ATP: ATP refers to the use of pacing stimulation techniques for termination of monomorphic ventricular tachyarrhythmias. Such techniques offer the potential for painless termination of VTs.
- Shock: High voltage shocks are given to restore VT/VF into sinus rhythm.
Different detection schemes and therapies can be programmed for different categories. For example, ATP may be appropriate treatment for slow VTs and shock therapy is an appropriate treatment for VF. After therapy is deliverd, the ICD monitors the next intervals to redetect sinus rate (which means the therapy worked) or redetect the arrhythmia (which results in resumed therapy).
Follow up
Like pacemaker implantation, the first 6-8 weeks after transvenous ICD implantation, patients are advised not to over-stretch their arm on the same side as the ICD (such as golf, swimming etc.) to allow time for the lead to mature.
Control visits for ICD are usually every 6 months. During this visit several electrical parameters are measured: battery status, sensing and pacing thresholds and impedance of all leads (see above).
Complications and inappropriate shocks
Also in ICDs problems of undersensing, oversensing, non-capture, changes in impedance, operative failures and long-term complications can occur (see above at pacemaker complications).
Additionally, a major complication in some patients with ICDs is the occurrence of inappropriate shocks. An inappropriate shock is shock therapy for anything else but ventricular fibrillation or ventricular tachycardia. This can be due to, for example, supraventricular tachycardia with fast ventricle response (including sinus tachycardia and atrial fibrillation), T-wave oversensing, detection of physiological- or other non-cardiac activity and lead- or device failure.
Cardiac resynchronisation therapy (CRT)
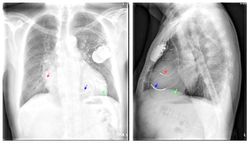
The red arrow is the right atrial lead
The blue arrow is the right ventricular lead
The green arrow is the coronary sinus lead
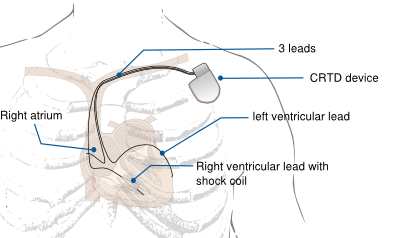
CRT-pacemaker (CRT-P) is a biventricular pacemaker with leads in both ventricles to ensure synchronized contraction. A CRT-defibrillator (CRT-D) is an ICD with biventricular pacing option. It appears that atrio-ventricular and intraventricular conduction delays further aggrevates left ventricular (LV) dysfunction in patients with underlying cardiomyopathies. Notably, left bundle branch block (LBBB) alters the sequence of LV contraction, causing wall segments to contract early or late. Dyssynchrony seems to represent a pathophysiological process that directly depresses ventricular function, causing LV remodelling and congestive heart failure and as a consequence causes a higher risk of morbidity and mortality. Atrio-biventricular pacing (CRT) for patients with symptomatic heart failure and intra- or interventricular conduction disturbances has proved beneficial.
Patients
CRT-P or CRT-D appears to be effective in patients with NYHA function class III/IV for congestive heart failure, LVEF =35%, QRS width = 120 ms (especially LBBB) who are on optimal medical therapy. Patients with NYHA function class IV should be ambulatory (no admissions for heart failure during the last month and a reasonable expectation of survival > 6 months).
Also patients with NYHA function class II, LVEF =35%, QRS width = 150 ms (especially LBBB) who are on optimal medical therapy are appropriate patients for CRT-D therapy.
Implantation
CRT-P or CRT-D implantion requires implantation of three transvenous leads:
- Right atrial lead
- Right ventricular lead
- Left ventricular lead. The left ventricular lead is usually positioned in the coronary sinus, alternatively it can be positioned epicardially on the left ventricle (by a surgical procedure) or intracardially in the left ventricle (through a transseptal puncture).
Programming, follow up and complications
Programming should specifically aim at ensuring atrial-synchronous permanent biventricular pacing, by performing AV-interval optimization (echocardiography guided or using invasive haemodynamic measurments) and performing ventricular-ventricle (VV) interval optimization. Further programming, follow up and complications are similar to pacemakers and ICDs (see above).
References
- Epstein et al. ACC/AHA/HRS Guidelines for Device-Based Therapy of Cardiac Rhythm Abnormalities. Heart Rhythm 2008;5:e1-62
- Vardas et al. Guidelines for cardiac pacing and cardiac resynchronization therapy: the task force for cardiac pacing and cardiac resynchronization therapy of the European society of cardiology. Developed in collaboration with the European Heart Rhythm Association. Eur Heart J 2007; 28:2256-95
- ECGpedia