Valvular Heart Disease
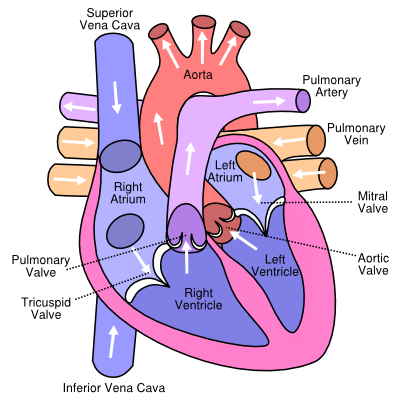
The four cardiac valves consist of either cusps or leaflets that close to prevent the blood from flowing backwards. When pressure behind the valve builds up, the valve opens, after blood has passed through, the pressure is reduced and the valve closes, actively or passively.
Epidemiology
Valvular heart diseases are a major burden to society and it is expected that the prevalence will increase.
Rheumatic valve disease used to be the most prevalent etiology of valvular cardiac diseases worldwide. Still, in developing countries, rheumatic heart disease remains the most common cause of valvular heart disease. Over the past 60 years, the etiology of most valvular heart diseases in industrialized countries shifted towards degenerative etiologies, mainly because of a decrease in acute rheumatic fever. However cardiac valve diseases remain common in industrialized countries, mainly because the decrease in rheumatic valve disease is compensated by an increase in degenerative valve disease, with an important contributing fact being the aging population of industrialized countries. The shift in pathologic etiology accounts for differences in patient characteristics and distribution of type of valvular lesions. SolerGalve
In the US population, the national prevalence of moderate and severe valve disease determined by echocardiography was estimated at 2,5%. In another cohort, prevalence based on clinical signs and symptoms, confirmed by echocardiographic imaging, the estimated prevalence of at least moderate valvular diseas was estimated at 1,8%. This difference indicates the under diagnosing of valvular heart disease, and illustrates the fact that diagnosis on the basis of clinical information alone is not reliable.Nkomo Prevalence did not change according to gender, but increased substantially with advancing age, with 13,2 % after the age of 75 years, versus <2% prior to 65 years old. The predominance of degenerative etiologies accounts for the higher prevalence in the elderly. The prevalence of degenerative valve disease is expected to rise with the aging population of Western countries.
Mitral regurgitation was found to be the most frequent valvular disease, with a prevalence of 1,7 %, followed by aortic regurgitation (0,5%), aortic stenosis (0,4%) and mitral stenosis (0,1%)
Mean age of patients presenting to the hospital in the Euro heart survey was 65 years.Iung In this survey, 63% of all cases of native valve disease were of degenerative aetiology. Aortic stenosis was found to be the most frequent valvular disease of patients referred for treatment. In 22% of all patients aetiology was rheumatic heart disease.
In developing countries, approximately 30 milion cases of rheumatic fever occur annually, in general before the age of 20.BurgeDeHoratius Approximately 60% of patients will develop rheumatic heart disease, which becomes clinically evident 1 to 3 decades later.CarapetisCurrieMathews Rheumatic heart disease remains the most common cause of valvular heart disease in third world countries. In western countries, rheumatic heart disease is the second most common cause of valvular heart disease
Pathophysiology
Normal valves |
| id=prxQ20XEEwg|autoplay=1|loop=0|right}} |
| This video shows a normal aortic valve on the left and mitral valve on the right. Video from the Visible Human Heart |
All cardiac valves have similar well defined interstitial cell layers, covered by endothelium. The three cell layers have specific features, and are named fibrose, spongiosa, and the ventricularis. During the cardiac cycle, the spongiosa rich in glycosaminoglycans, facilitates the relative rearrangements of collagenous and elastic layers. Valvular interstitial cells (VIC) are abundant in all layers of the cardiac valves and comprise a diverse, dynamic population of resident cells. Regulation of collagen and other matrix components is ensured by enzymes, synthesized by VICs. Integrity of valvular tissue is maintained by interaction of valvular endothelial cells (VECs) with VICs. Changes and remodeling of valvular interstitial and endothelium cell leads to changes in properties of the valve and potentially also valve function.
Aortic valve
The tricuspid aortic valve separates the left ventricle outflow tract from the aorta. Behind the three semilunar shaped cusps of the aortic valve are dilated pockets of the aortic root, called sinuses of Valsalva. The right coronary sinus gives rise to the right coronary artery, the left coronary sinus gives rise to the left coronary artery. The commissures are the areas where attachments of two adjacent cusps to the aorta meet.
The commissure between the left en non coronary leaflets is positioned along the area of mitro-aortic continuity. The three cusps ascend towards the commissures and descend to the basal attachment with the aorta. Opening and closure of the aortic valve is a passive, pressure driven mechanism in contrast to the mitral valve. Tissue of the aortic cusps is stretched via backpressure in diastolic phase with elongation and stretching of elastin. In the systolic phase, recoil of elastin ensures relaxation and shortening of the cuspal tissue.Rajamannan Optimal functioning of the valve requires perfect alignment of the three cusps.
Mitral Valve
The mitral valve was named after a Mitre, by Andreas Vesalius (De Humani Corporis Fabrica, 1543).Di This active valve is located at the junction of the left atrium and left ventricle. The mitral valve apparatus contains five functional components; leaflets, annulus, chordae tendineae, papillary muscles and subajacent myocardium. The annulus is a junctional zone of discontinuous fibrous and muscular tissue that joins the left atrium and ventricle. The anterior leaflet spans about one third of the primary fibrous, anterior part of the annulus. Part of the mitral valve anterior leaflet is in direct fibrous continuity with the aortic valve annulus, the mitro-aortic continuity. The posterior, ventricular leaflet is attached to the posterior predominantly muscular half to two third of the annulus. Due to the asymmetric leaflets, the mitral valve orifice has a funnel shape.
Chordae tendinae from both the anterior and posterior papillary muscles are attached to each leaflet. The papillary muscles contract and pull the chordae tendinae during systole, which closes the two mitral valve leaflets.
The mitral valvular complex comprises the mitral valve apparatus and left atrial en ventricular myocardium, endocardium and the mitro-aortic continuity. It contributes to the formation of the left ventricular outflow tract. The timed passage of blood through the valve as well as the tight closure during systole is facilitated by combined actions of the mitral valvular complex.Muresian
Pulmonary Valve
The structure of the pulmonary valve is analogous to the aortic valve structure. The leaflets are semilunar shaped, with semilunar attachments. The pulmonary valve has no traditional annulus. Anatomically, three rings can be distinguished, superior at the sinotubular junction, at the musculoarterial junction and a third ring at the base of the sinuses.MillWilcoxAnderson
Tricuspid valve
The tricuspid valve is located at the junction between the right atrium and right ventricle. The tricuspid valve apparatus consists of 3 leaflets, chordae tendinae, an anterior, posterior and often a third papillary muscle. The peripheral ends of the septal, anterosuperior and inferior or mural leaflets are referred to as commissures. The tricuspid valve has no well defined collagenous annulus. The three leaflets are attached to a fibrous elliptic shaped annulus. The direct attachment of the septal leaflet is a distinctive feature of the tricuspid valve. The prominent papillary muscles support the leaflets at the commissures.
The anterior papillary muscle provides chords to the anterior and mural leaflets, the posterior papillary muscle provides chords to the mural and septal leaflets.
Normal valve function requires structural integrity and coordinated interactions among multiple anatomic components.PaderaSchoen A variety of pathophysiologic mechanisms can cause cardiac valve disease.
Valvular stenosis, defined as inhibition of forward flow secondary to obstruction caused by failure of a valve to open completely, is almost always caused by a primary cuspal abnormality and a chronic disease process.
Valvular insufficiency, defined as reverse flow caused by failure of a valve to close completely, may result from either intrinsic disease of the valve cusps or from damage to or distortion of supporting structures without primary cuspal pathology
Rheumatic valve disease
Chronic rheumatic valve disease is characterized by chronic, progressive deforming valvular disease. Anatomic lesions combine to varying degrees fibrous, or fibrocalcific distortion of leaflets or cusps, valve commissures and chordae tendineae, with or without annular or papillary muscle deformities.
Stenosis results from fibrous leaflet and chordal thickening and commissural and chordal fusion with or without secondary calcification. Fusion of a commisure in an open position can cause regurgitation, as well as scarring induced retraction of chordae and leaflets.
Aortic valve Stenosis
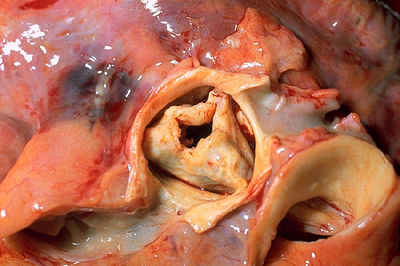
Obstruction of the left ventricle outflow can occur at subvalvular level (eg hypertrophic cardiomyopathy), supravalvular level or valvular level. Aortic valve stenosis is left ventricle outflow obstruction at valvular level.
In industrialized countries, aortic stenosis is the most common lesion among patients referred for treatment of valvular disease.IungB Age-related degenerative calcified aortic stenosis is the most common cause of aortic stenosis in adults in North America and Western Europe. The second most common cause is calcification of a congenitally bicuspid aortic valve. Other rare causes of calcified aortic stenosis include Fabry disease, lupus erythematosus, Paget disease, and ochronosis with alkaptonuria. The most common etiology of aortic stenosis worldwide remains rheumatic heart disease.
Prevalence of aortic valve abnormalities increases due to age-related pathology in the ageing population.
The first detectable macroscopic modifications of the calcification process is named aortic valve sclerosis. Rajamannan Aortic sclerosis, seen as calcification or focal leaflet thickening with normal valve function, was detected in 25% of people at 65 years of age, this increases to 48% in people aged >75% in a population-based echocardiographic study.Otto Otto2
The prevalence of calcified aortic stenosis is estimated at 2 % of people 65 years of age, increasing to 3-9% after the age of 80 years.NkomoOtto2
Calcified degenerative aortic valve stenosis was previously considered to be the result of a passive degenerative process due to longterm mechanical stress in combination with calcium accumulation. Recently this concept is revised. Calcified degenerative aortic stenosis is considered an active pathobiological process, including proliferative and inflammatory changes, lipid accumulation, renin-angiotensin system activation, valular interstitial cell transformation, ultimately resulting in calcification of the aortic valve.RajamannanGershBonowRajamannan2 OBrien Mohler Risk factors for development of calcific aortic stenosis are similar to those for vascular atherosclerosis such as diabetes, hypertension, and cholesterol levels.Stewart Stritzke Progressive calcification leads to immobilization of the cusps causing stenosis.
Severity of outflow obstruction gradually increases in aortic valve stenosis. Left ventricular output is maintained by adaptation of the increasingly hypertrophic left ventricle. This compensational mechanism serves to normalize the left ventricle wall stress. Left ventricular hypertrophy in combination with the prolonged systolic phase of the cardiac cycle results in increased myocardial oxygen demand. The mismatch between oxygen demand and supply is the main mechanism for angina in aortic stenosis.
As the stenosis progresses, the left ventricle becomes less compliant with subsequent limited preload reserve. Eventually, the left ventricle will decompensate with a decline in cardiac output and rise in pulmonary artery pressure.
Aortic stenosis is assessed by estimating the mean systolic pressure gradient and aortic valve area (AVA). The normal aortic valve area is 3-4 cm2. Mild aortic stenosis is defined as an aortic valve area 1.5 cm2, mean gradient less than 25 mm Hg, or jet velocity less than 3.0 m per second, moderate aortic stenosis as an area of 1.0 to 1.5 cm2, mean gradient 25 to 40 mmHg, or jet velocity 3.0 to 4.0 m per second. A valve area of <1 cm2, a mean gradient greater than 40 mm Hg, or jet velocity greater than 4.0 m per second implies severe aortic stenosis The valve area may decrease by as much as 0.12 ± 0.19cm2 per year.OttoBurwaskLegget In late stages of severe aortic stenosis, cardiac output declines due to systolic dysfunction of the left ventricle, with a decline in the transvalvular gradient.
| Aortic stenosis severity | Valve area | Mean pressure gradient | Jet velocity |
|---|---|---|---|
| Mild | area 1.5 cm2 | less than 25 mm Hg | less than 3.0 m per second |
| Moderate | area 1.0 to 1.5 cm2 | 25 to 40 mmHg | 3.0 to 4.0 m per second |
| Severe | area less than 1.0 cm2 | greater than 40 mm Hg | greater than 4.0 m per second |
Clinical Presentation
Symptoms of degenerative aortic stenosis manifest with progression of the disease. The first symptoms usually commence in the seventh or eight decade. Symptoms are typically noted on exertion. Dyspnoea on exertion is the most common encountered first symptom. Other symptoms are angina, precipitated by exertion and relieved by rest, syncope and heart failure. The findings on physical examination vary with the severity of the disease. On auscultation, a systolic ejection crescendo-decrescendo murmur, radiating to the neck is audible, often accompanied by a thrill. An elevated left ventricular pressure in patients with aortic stenosis, in conjunction with mitral annulus calcifications predisposes to rupture of mitral chordae tendineae, which may produce a regurgitant systolic murmur.Brener Mihaljevic
The first heart sound is usually normal or soft in patients with aortic stenosis. The second heart sound may be delayed due to prolongation of systolic ejection time. The S2 also may be single because of superimposed aortic and pulmonic valve components, or the aortic valve component is absent or soft because the aortic valve is too calcified and has become immobile. If the aortic component is audible, this may give rise to a paradoxical splitting of S2. A pronounced atrial contraction can give rise to a palpable and audible S4.
When stroke volume and systolic pulse pressures fall in severe aortic stenosis, a pulsus parvus (small pulse) may be present. A wide pulse pressure is also characteristic of aortic stenosis. A pulsus parvus et tardus (the arterial pulse is slow to increase and has a reduced peak) can be appreciated by palpating the carotid pulse of patients with severe aortic stenosis. The stenotic valve decreases the amplitude and delays the timing of the carotid upstroke. Rigidity of the vasculature may hamper this sign in the elderly.
Diagnostic options
Chest Radiography
In aortic stenosis, cardiac silhouette and pulmonary vascular distribution are normal unless cardiac decompensation is present. Post-stenotic dilatation of the ascending aorta is frequent. Calcification of the valve is found in almost all adults with severe aortic stenosis; however, fluoroscopy may be necessary to detect it. A late feature in patients with aortic valve stenosis is cardiomegaly. In patients with heart failure, the heart is enlarged, with congestion of pulmonary vasculature.
Electrocardiography
In approximately 85% of patients with aortic stenosis, left ventricle hypertrophy, with or without repolarization abnormalities is seen on electrocardiography (ECG). Left atrial enlargement, left axis deviation and conduction disorders are also common. Atrial fibrillation can be seen at late state and in older patients or those with hypertension.
Echocardiography |
The best non-invasive diagnostic tool to confirm the diagnosis of aortic stenosis, assess the number of cusps and the annular size, is ultrasonic examination of the heart. Quantification of valvular calcification is possible. In 1998, the American college of cardiology/American Heart Association (ACC/AHA) task force Bonow recommended the diagnostic use of echocardiography.
Echocardiographic imaging evaluates the severity and etiology of the primary valvular lesion, secondary lesions, and coexisting abnormalities. The size and function of the atria and ventricles can be evaluated as well as hemodynamic characteristics. Echocardiography is also performed for postprocedural evaluation of patients. Transthoracic echocardiography is recommended for re-evaluation of asymptomatic patients: every year for severe AS; every 1 to 2 years for moderate AS; and every 3 to 5 years for mild AS.Bonow2
To assess the severity of aortic stenosis, transvalvular gradients and maximum jet velocity is measured using Doppler echocardiography, and aortic valve area is calculated. The systolic gradient across the stenotic aortic valve depends on stroke volume, systolic ejection period, and systolic pressure in the ascending aorta. The stenotic valve area is inversely related to the square root of the mean systolic gradient. Due to their flow-dependency these measurements are most valuable in normotensive patients.
Valve thickening and calcification, as well as reduced leaflet motion can also be assessed using Doppler.
Computed tomography |
Although the role of computed tomography (CT) in clinical management is currently not well defined, this imaging modality could improve assessment of the ascending aorta. CT has an established role in evaluating the presence and severity of aortic root and ascending aortic dilatation in patients with associated aortic aneurysms. The high sensitivity and specificity of CT in detecting high-grade coronary artery stenosis could be useful to preoperatively rule out coronary artery disease.
Both electron beam and multislice cardiac CT can be useful in quantifying valve calcification, which have been shown to correlate with echocardiographic assessment and clinical outcome. Prior to transcatheter aortic valve implantations, CT provides information concerning the aortic valve area, annulus size, and the distance between the aortic cusps and the coronary ostia.
Cardiac Magnetic Resonance Imaging |
Cardiac MRI (CMR) has an established role in evaluating aortic root and ascending aorta anatomy. It can be used to measure the aortic valve area, but the role of CMR in the management of aortic stenosis is currently not well defined.
Cardiac Catheterization |
Cardiac catheterization remains the gold standard to detect coronary artery disease. Currently, in patients with aortic stenosis, cardiac catheterization is most often performed to identify the presence of concomitant coronary artery disease (CAD). In patients with inconclusive noninvasive tests, hemodynamic abnormalities can be assessed by cardiac catheterization. Coronary angiography is recommended prior to aortic valve replacement.
Exercise Testing |
Since aortic stenosis is a progressive disease, most common in the elderly population, many patients with aortic stenosis do not recognize gradually developing symptoms and cannot differentiate fatigue and dyspnea from aging and physical deconditioning. Lifestyle modification may mask symptoms. Although contraindicated in patients with severe aortic stenosis, Exercise testing is useful for risk stratification and eliciting symptoms. Under supervision, it is reasonable to propose exercise testing in patients >70 years who are still highly active.
Treatment
Medical treatment |
For many years the standard of care for patients with significant aortic valve stenosis has been to provide antibiotic prophylaxis against infective endocarditis. However, current AHA guidelines for prevention of infective endocarditis no longer recommend antibiotic prophylaxis for this group of patients. Exceptions are patients with a prior episode of endocarditis, patients with prosthetic valves or with additional complex cardiac lesions with a high risk for the development of endocarditis. Patients who have had rheumatic fever should still receive antibiotic prophylaxis against recurrences of rheumatic fever
No medical treatment has proven to delay the progression of aortic stenosis. Surgery is inevitable for symptomatic patients. Patients at prohibitive risk for intervention may benefit from medical treatment including digitalis, diuretics, ACE inhibitors, or angiotensin receptor blockers, if experiencing heart failure. Beta-blockers should be avoided in these circumstances.
| Current Guidelines |
|---|
| Indications for aortic valve replacement: |
Class I
|
| Class IIa
AVR is reasonable for patients with moderate AS undergoing CABG or surgery on the aorta or other heart valves (Level of Evidence: B) |
Class IIb
|
| Class III
AVR is not useful for the prevention of sudden death in asymptomatic patients with AS who have none of the findings listed under the Class IIa/IIb recommendations. (Level of Evidence: B) |
Surgery |
The infinitive treatment for aortic valve stenosis is aortic valve replacement.
The first cardiac valve surgery under direct vision was an aortic valve replacement, performed in 1960 by dr. Dwight Harken.Stephenson The aortic valve was replaced by a caged ball valve, which became the standard for aortic valve replacement.Emery Chaikof
A total of more than 70 different mechanical aortic valve models have been introduced in aortic valve replacement and implanted in humans in the past 5 decades. The mechanical prostheses can be divided into 3 large groups: the first generation of ball valves, second generation of tilting-disc valves, and the last generation of bileaflet valves.Vitale Mechanical prosthesis are extremely durable but require continuous use of anticoagulants.
Biological valves include homografts and autografts, as well as stented bioprostheses. Stented bioprostheses are constructed of porcine valves or bovine pericardium sewn onto an artificial stent. All heterograft valvs are preserved with glutaraldehyde, to reduce the antigenicity of the tissue and prevent calcification. The fixation process can be performed at various pressures. Higher fixation pressures may lead to earlier calcification. First-generation bioprostheses were porcine valves, preserved with high-pressure fixation (60 to 80 mmHg) and placed in the annular position
Second-generation prostheses are of porcine or pericardial origin, and are treated with low- pressure (0.1 to 2 mmHg)or zero-pressure fixation. Several second-generation bioprostheses may be placed in the supra-annular position, which allows implantation of a slightly larger prosthesis. Third-generation prostheses are treated with zero- or low-pressure fixation and additional processes to reduce calcification.
In 1962 Donald Ross implanted the first aortic valve allograft. In 1967 he replaced a patient’s malfunctioning aortic valve with the patients own pulmonary valve. An aortic or pulmonary valve homograft was then used to replace the patient’s pulmonary valve. This procedure is known as the Ross Procedure. Currently, the Ross procedure may be considered for bicuspid aortic valve stenosis, in particular for young women of reproductive age.
Transcatheter intervention |
In 2002, the first transcatheter aortic valve implantation was performed by Dr. Alain Cribier Cribier. A transcatheter aortic valve implantation is a less invasive treatment option for patients at prohibitive risk for conventional aortic valve replacement. In this technique, the native valve is not excised. After balloon valvuloplasty, the prosthetic valve is implanted in the aortic position, with the frame of the prosthesis covering the native valve. The bioprosthesis can be implanted retrograde or antegrade. Currently 4 different approaches may be used in this technique. (table…). Transcatheter aortic valve implantation is assessed in randomized clinical trials and registries.
| The current 4 different approaches are: |
|---|
| Transfemoral, retrograde
Transapical, antegrade Transaortic, retrograde Transsubclavian, retrograde |
Prognosis
Aortic valve stenosis has a severe prognosis when any symptoms are present, with survival rates of only 15–50% at 5 years. Strongest predictors of poor outcome in the elderly population are high New York Heart Association (NYHA) class (III/IV), associated mitral regurgitation and left ventricular dysfunction. Survival is only 30% at 3 years with the combination of these three factors.
Bicuspid Aortic valve
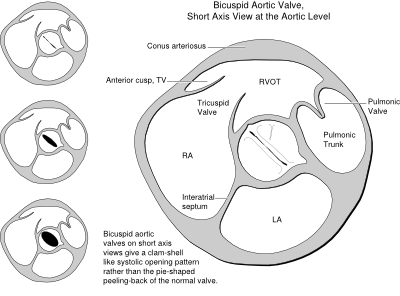
Bicuspid Aortic valve disease affects as many as 1-2% of the population, and is the most frequent congenital cardiovascular malformation in humans.Fedak
A bicuspid aortic valve may be part of a phenotypic continuum of congenital aortic valve disorders, associated with unicuspid valves, bicuspid valves, the normal tricuspid valves and the rare quadricuspid forms. Understanding of the pathogenesis of aortic valve malformation remains incomplete.
Bicuspid aortic valves are three to four times more common in men than in women. Bicuspid aortic valve disease results from abnormal cusp formation during valvulogenesis, but coexisting genetic abnormalities of the aorta and proximal coronary vasculature are often present. Moreover, nonvalvular findings occur in up to 50% of patients with bicuspid aortic valves. Associated findings are aortic dilation, aneurysms and dissection.
During valvulogenesis, adjacent cusps of the bicuspid valve fuse to form a single aberrant cusp. This fusion results in large leaflet, yet smaller than 2 normal cusps, with most often a central raphe or ridge. Fusion of the right coronary and noncoronary cusps is associated with cuspal pathology. Fusion of the right and left coronary cusps is associated with coarctation of the aorta.
Although endocarditis can be a devastating complication of bicuspid aortic valve disease, straightforward bicuspid aortic valve disease is no longer an indication for bacterial endocarditis prophylaxis according to the ACC/AHA practice guidelines. The risk of endocarditis is felt to be low in patients with straightforward bicuspid aortic valve disease. An exception to this recommendation is a patient with a prior history of endocarditis.Nishimura
Clinical Presentation |
In infancy, bicuspid aortic valve disease is often asymptomatic. By adolescence an estimate 1 of 50 children born with these abnormalities will have clinically significant obstruction or regurgitation.Bonow2
Complications of bicuspid aortic valve disease are common in adulthood.Tzemos The abnormal shear stress leads to valve calcification and further aortic root dilation has been reported. Bonow3 The most common complication is aortic stenosis, caused by premature fibrosis, stiffening, and calcium deposition. The majority of patients under 65 years of age with significant aortic valve stenosis have bicuspid aortic valve disease. A more rare complication of bicuspid aortic valve disease is aortic regurgitation. 15% of all cases of aortic regurgitation in the Euro Heart survey had bicuspid aortic valve disease. On auscultation, an ejection sound can be audible, best heard at the apex. There may be associated murmurs of aortic stenosis, incompetence, or coarctation of the aorta when these lesions are present.
Diagnostic options |
Echocardiography
Echocardiography is used to confirm the diagnosis of bicuspid aortic valve disease. Reported sensitivities and specificities of echocardiography for detecting BAV anatomy are 92% and 96% respectively. To establish the diagnosis, visualization of the aortic valve in systole in the short-axis view is essential. During diastole, the raphe can make the valve appear trileaflet. In the long-axis view, the valve often has an eccentric closure line and there is doming of the leaflets. Transesophageal echocardiography may improve visualization of the leaflets in case of inconclusive transthoracic echocardiography.
In all patients, serial transthoracic echocardiography should be performed to evaluate the valve and disease progression. Annual cardiac imaging is recommended for patients with significant valve lesions or with aortic root diameters >40 mm. Complete imaging of the thoracic aorta should be performed periodically for surveillance.SiuSilversides
Cardiac Magnetic Resonance Imaging and Computed Tomography
The thoracic aorta is visualized by alternative imaging modalities such as cardiac magnetic resonance imaging (MRI) or computer tomography (CT). Both cardiac MRI and CT images can help to confirm the bicuspid anatomy of the aortic valve.
Treatment |
Medical treatment
In patients with bicuspid aortic valve disease, high blood pressure should be treated aggressively. The ACC/AHA guidelines for the management of adult congenital heart disease and guidelines for the management of patients with valvular heart disease suggest that it is reasonable to use beta-blockers in this population (Class IIa recommendation).Warnes This is in accordance with the standard of care at many centers to slow the progression in Marfan-associated aortopathy.
Surgery
Indications for surgery are similar to indications for patients with “degenerative aortic valve disease”; intervention is indicated for severe valvular dysfunction, symptomatic patients, and patients with evidence of abnormal left ventricular dimensions and function.
In children and young adults, the bicuspid valve is not calcified and balloon valvuloplasty is recommended. A prosthetic valve implantation would be suboptimal due to the continuing growth of the child.
Indications for valvuloplasty in children include peak-to peak gradients >50 mm Hg with ST- or T-wave changes at rest or with exercise. Valvuloplasty is also indicated for symptomatic children with peak-to-peak gradients >60 mm Hg. Warnes
Surgical options for adult bicuspic aortic valve disease include valve replacement (bioprosthetic or mechanical valves), Ross procedure or valve repair (for those with aortic incompetence) Surgical aortic valve replacement is the most common procedure in adults with bicuspid aortic valve disease, for either aortic valve stenosis or regurgitation. Indications of interventions are similar to those described for tricuspid aortic valve disease in the ACC/AHA guidelines for the management of patients with valvular heart disease.Bonow3
Approximately 30% of adults with bicuspid aortic valve disease undergoing aortic valve replacement will need aortic root surgery. Surgical attention for dimensions of the aortic root is essential because of the risk for further root dilation. The ascending aorta in patients with bicuspid aortic valve disease increases 0.2 to 1.2 mm/year.Thanassoulis
Guidelines suggest that changes in root size more than 0.5 cm/year are an indication for root replacement. Aortic root dimensions of 5.0 cm require intervention and aortic root dimensions of 4.5 cm require intervention if surgery is performed for valvular indications according to current guidelines.
Prognosis |
Life expectancy in adult patients with bicuspid aortic valve disease is not shortened when compared to the general population. 10-year survival in asymptomatic adults with bicuspid aortic valve disease with a spectrum of valve function, was 96%.Tzemos In asymptomatic adults with bicuspid aortic valve disease without significant valve dysfunction the 20-year survival was 90%.Michelena
Aortic Regurgitation
A variety of aetiologies can cause aortic regurgitation by preventing proper coaptation of the aortic valve leaflets with a subsequent diastolic reflux of blood from the aorta into the left ventricle. Etiology of aortic regurgitation can be primary valvular, or it can be primarily caused by aortic root or disease.
The origin of primary valve disease may be calcific aortic disease, idiopathic degenerative disease, endocarditis, rheumatic disease, a biscuspid aortic valve, or myxomatous proliferation of valvular tissue. In the majority of patients the disease is caused by rheumatic disease. However, in Western countries the disease is most often of degenerative origin. In the Euro Heart Survey degenerative aortic regurgitation accounted for approximately half of the cases of aortic regurgitation, 15% of cases had a bicuspid aortic valve.IungB Accelerated degeneration of valve leaflets, resulting in regurgitation similar carcinoid syndrome related regurgitation, can be caused by certain anorectic medications, such as fenfluramine and phentermine.Mihaljevic
Aortic annulus dilation, without primary involvement of the leaflets may result in aortic regurgitation due to leaflet separation. Aortic regurgitation of primary aortic root or annulus aetiology includes idiopathic aortic root dilatation, aortic dissection, trauma, and chronic severe systemic hypertension. Aortitis represents less than 5% of the aetiologies of aortic regurgitation and may be due to inflammatory disease, such as giant cell, Takayasu and Behcet syndrome. Syphilis and ankylosing spondylitis may affect the aortic valve, but may also be associated with aortic dilatation. Other systemic arteritides and connective tissue disorders such as Marfan syndrome, Reiter disease, Ehlers-Danlos syndrome, osteogenesis imperfecta, and rheumatoid arthritis can lead to annular dilatation and valvular insufficiency. In patients without generalized tissue disease the same pattern of ascending aortic enlargement is known as annuloaortic ectasia. Chronic aortic regurgitation itself may lead to progressive aortic root dilatation.
Aortic valve regurgitation is often accompanied by other valvular abnormalities. Aortic valvular insufficiency is most commonly seen in combination with aortic stenosis. Aortic insufficiency due to rheumatic aetiology is often associated with mitral valve disease. The valve leaflets are retracted by fused commissures and by fibrotic scarring of the leaflets itself.
Aortic cusp prolapse can be isolated, or due to myxomatous degeneration, sometimes with associated mitral or tricuspid valve involvement. In 15% of patients with ventricular septum defect, prolapse of an aortic cusp leads to aortic insufficiency.
Isolated aortic regurgitation is often caused by a primary aortic annular etiology. Rheumatic origin is much less common in patients with pure aortic regurgitation. In approximately 10% of cases, aortic regurgitation results from infective endocarditis, with perforation or erosion of leaflets.
It is of considerable clinical importance to distinguish between acute aortic regurgitation and chronic regurgitation since acute aortic regurgitation can be life-threatening if not treated immediately, in contrast to chronic regurgitation which can be tolerated for years.
Pathophysiology |
In patients with aortic insufficiency the regurgitating volume increases the total stroke volume. This volume might equal the effective forward stroke volume in patients with severe aortic regurgitation. In chronic aortic regurgitation, several compensatory mechanisms ensure cardiac output.
Acute aortic regurgitation
Acute aortic regurgitation is a sudden hemodynamically significant aortic incompetence, which can often be catastrophic. An increase in left ventricular end diastolic volume with absence of ventricular remodelling may lead to elevated left atrial and pulmonary artery wedge pressure and decreased effective cardiac output, with compensatory tachycardia to maintain sufficient output.
Acute severe aortic regurgitation typically occurs with infective endocarditis, trauma, and aortic dissection. The left ventricle cannot dilate sufficiently. The patient may present with chest pain due to increased myocardical oxygen consumption combined with decreased coronary bloodflow from changes in diastolic perfusion. Coronary bloodflow in acute aortic insufficiency occurs during stystolic cardiac phase. Other symptoms of acute aortic regurgitation are tachypnea, tachycardia and rapidly progressive pulmonary edema and/or cardiogenic shock.
Chronic Aortic regurgitation
Aortic root dilatation, annular dilation and congenital bicuspid valve are, in developed countries, the most common causes of severe chronic aortic valve regurgitation.
The slow process of chronic aortic regurgitation allows adaptation of the ventricle to the increased preload and afterload. The left ventricular compensates to the regurgitant flow, the increased volume and pressure by enlargement. The left ventricle end diastolic pressure remains relatively low and does not approach the aortic diastolic pressure. The additional stroke volume is responsible for increased systolic pressure and eventually the wide pulse pressure. The systolic hypertension further increases left ventricle afterload.
In contrast to the compensatory mechanism in mitral valve regurgitation, a modest concentric left ventricular hypertrophy accompanies the eccentric hypertrophy, with a normal mass-to-volume ratio.FeiringRumberger In a chronic state, progressive left ventricle dilatation leads to pre- and afterload mismatch. With gradually decompensation and deterioration of systolic function, the ventricle is not able to sustain perfusion.
| Causes of chronic aortic regurgitation |
|---|
| Aortic root/annular dilation
Congenital bicuspid valve Previous infective endocarditis Rheumatic In association with other diseases |
Clinical presentation |
Patients with aortic regurgitation typically present with symptoms of left sided heart failure including dyspnea on exertion, orthopnea, fatigue, and occasionally paroxysmal nocturnal dyspnea. Angina is less common in patients with aortic regurgitation compared to aortic stenosis. The reduced aortic diastolic blood pressure reduces the coronary blood flow, resulting in angina. The same mechanism is presumed to cause syncope.
Diagnostics |
Chest Radiography
Chest Radiography in acute aortic regurgitation reveals minimal cardiac enlargement, with normal aortic root and arch. In acute aortic regurgitation, signs of left heart failure are frequent. Cardiomegaly with left ventricular enlargement is the main feature on chest radiography in chronic aorta regurgitation. The ascending aorta may be enlarged in case of an aortic aneurysm or aortic dissection but Chest X-ray is not a sensitive examination to detect ascending aortic aneurysm.
Pulmonary congestion is noted at advanced stages of chronic AR, when heart failure has developed.
Electrocardiography
Electrocardiography in patients with aortic regurgitation may be normal early in the disease. Left ventricular hypertrophy is the main feature of aortic regurgitation, with or without associated repolarization abnormalities. Left axis deviation may be present.
Echocardiography
Echocardiography is used to evaluate the anatomy of the aortic valve and other valves, as well as aortic leaflets and the aortic root. The regurgitation mechanism and aetiology can be assessed. Three types of mechanisms can be identified; enlargement of aortic root with normal cusps, cusp prolapse or fenestration and poor cusp tissue quality or quantity. Doppler is used for quantifying the aortic regurgitation by the width of regurgitant jet and its extension into the LV, the rate of decline of aortic regurgitant flow and diastolic flow reversal in the descending aorta.
Severe aortic regurgitation is defined as effective regurgitant orifice (ERO) area of >0.30cm2, regurgitant volume >60mL, or a regurgitant fraction of >50%
Preoperatively transoesophageal echocardiography is performed to more accurately evaluate the anatomy and mechanism of the aortic valve regurgitation
Treatment |
The only direct method to reduce aortic regurgitation is surgical treatment. However, some patients may benefit from medical treatment.
Medical treatment
The relative reduction of myocardial blood supply due to increased demand and/or associated obstructive coronary artery disease may cause angina. Angina may be treated by reducing aortic regurgitation, reduction of myocardial demand of revascularization of the myocardium. Clinical heart failure is treated with traditional therapy, including digitalis, diuretics, and ACEI. In severe heart failure, parenteral inotropic and vasodilator therapy may be needed.
| Current Guidelines: Medical treatment of Aortic Regurgitation |
|---|
| Class I
Vasodilator therapy is indicated for chronic therapy in patients with severe AR who have symptoms or LV dysfunction when surgery is not recommended because of additional cardiac or noncardiac factors. (Level of Evidence: B) |
| Class IIa
Vasodilator therapy is reasonable for short-term therapy to improve the hemodynamic profile of patients with severe heart failure symptoms and severe LV dysfunction before proceeding with AVR. (Level of Evidence: C) |
| Class IIb
Vasodilator therapy may be considered for long-term therapy in asymptomatic patients with severe AR who have LV dilatation but normal systolic function. (Level of Evidence: B) |
Class III
|
Surgical treatment
Surgical treatment in case of isolated Aortic valve regurgitation is aortic valve replacement. Aortic valve replacement is indicated in all symptomatic patients with severe Aortic regurgitation and in all asymptomatic patients with severe Aortic regurgitation with left ventricular ejection fraction (LVEF) <50% or left ventricular dilatation (end-diastolic diameter >75 mm or end-systolic diameter >55 mm).
When an aneurysm of the aortic root is associated with severe aortic regurgitation a Bentall procedure can be indicated. The ascending aorta is replacement by a composite graft comprising an aortic prosthesis, with re-implantation of the coronary arteries. When aortic enlargement is localized in the supra-coronary part of ascending aorta, replacing the supra-coronary section of the ascending aorta may be sufficient which is considered technically easier.
Although the prosthetic valve replacement remains the standard for aortic valve regurgitation, aortic valve repair procedures are performed with a combination of different surgical techniques. The quality of the cusps is essential for repair. The annulus and sinotubular junction can be surgically readapted to the cusps, eliminating the regurgitation.
| Current Guidelines |
|---|
Class I
|
Class IIb
|
Mitral Stenosis
Etiology and pathology |
The leading cause of mitral stenosis is rheumatic fever, causing postrheumatic deformities. Other rare aetiologies of left atrial outflow obstruction include congenital and degenerative mitral valve stenosis, severe mitral annular and/or leaflet calcification carcinoid disease, neoplasm, left atrial thrombus, infective endocarditis with large vegetations, certain inherited metabolic disorders such as Fabry’s disease, mucopolysaccharidosis, Whipple’s disease, gout, rheumatic arthritis, lupus erythematosus, methysergide therapy, and cases related to previous implanted prosthesis or commisurotomy.
Rheumatic valvular disease causes diffuse thickening of the valve leaflets by fibrous, or fibrocalcific distortion, with fusion of one or more commisures valve commisures, and fusion and shortening of the subvalvular apparatus. This combined with increasingly rigid cusps results in narrowing of the valve.
The area of the normal mitral valve orifice is 4-6 cm2. In patients with mitral stenosis, when the valve area approaches 2 cm2 or less, an early, mid and late diastolic transvalvular gradient is present between the left atrium and ventricle. With progressive mitral stenosis, transvalvular pressure gradient increases. Mitral transvalvular flow depends on cardiac output and heart rate. Shortening of diastolic phase in increased heart rate causes symptoms by reducing forward cardiac output. Mitral stenosis develops gradually, and may be asymptomatic for years.
Clinical presentation |
Patients with mitral stenosis may be asymptomatic for years. Mean age of presentation of symptoms is fifty to sixty years old. The presenting symptom in patients with mild mitral stenosis is typically dyspnea precipitated by stress or atrial fibrillation. Progression of disease with increasing left atrial and pulmonary venous pressures will cause progressive dyspnea. At advanced stage, patients are often thin and frail and complain of weakness and fatigue due to low cardiac output. When pulmonary hypertension and right ventricular failure develop, signs of tricuspid regurgitation, abdominal discomfort due to hepatomegaly and ascites can be present.
On physical examination, heart size is usually normal. An apical diastolic thrill can be palpated. A holodiastolic murmur can be auscultated at the apex with a presystolic accentuation in sinus rhythm. The intensity of this murmur is determined by the transmitral gradient. In patients with severe stenosis, calcified leaflets, or low cardiac output no diastolic murmur may be audible. An opining snap may be present due to sudden tensing of the pliable leaflets during opening. S1 may be loud when the mitral leaflets are pliable. Thickening and calcification will diminish S1 in more advanced stages. With pulmonary hypertension, S2 becomes prominent and a murmur of tricuspid regurgitation located at the xyphoid can be present. In severe mitral valve stenosis with pulmonary hypertension, pulmonary rales are audible. Intermittent malar flushes, jugular distension, and peripheral cyanosis may be present
Diagnostics
Chest Radiography
Signs of left atrial enlargement are often the earliest changes on chest radiography.
Straightening of the left heart border by prominent pulmonary arteries coupled with left atrial enlargement can be observed as well as a double contour of the left atrium and elevation of the left main stem bronchus. Distension of the pulmonary arteries and veins in the upper lung fields and pleural effusions indicate elevated pulmonary pressures. Kerley B lines may be observed in severe mitral stenosis.
Electrocardiography
Electrocardiography is in many cases normal and cannot asses the severity of mitral stenosis accurately. Atrial arrhythmias are more common in patients with advanced mitral stenosis. In sinus rythm signs of left atrial enlargement may be present with a prolonged P wave and a negative deflection in lead V1 and left axial deviation of P wave. Signs of right ventricular hypertrophy with right-axis deviation, a tall R wave in V1, and secondary ST-T-wave changes may be present in cases of severe pulmonary hypertension
Echocardiography
The primary diagnostic method for assessing mitral valve pathology and pathophysiology is echocardiography. The severity and consequences of the mitral stenosis can be evaluated. The valvular anatomy and morphology including valve thickening, mobility, calcification, subvalvular deformity and anatomic lesions can be assessed
Rheumatic mitral stenosis can cause reduced diastolic excursion of the leaflets and thickening or calcification of the valvular and subvalvular apparatus. Doppler is used to determine the peak and mean transvalvular pressure gradients. The valve area can be measured accurately by three-dimensional echocardiogram. Transesophageal echocardiography is able to provide more detailed information of the mitral valvular pathology than transthoracic echocardiography.
Cardiac Catheterization
Cardiac catheterization can provide information regarding coronary artery status. This diagnostic tool is not necessary to establish the diagnosis of mitral stenosis.
Prognosis
The progression of mitral stenosis ranges from 0.1–0.3cm2/year.VahanianIung Survival rates of 80% at 10 years have been reported with asymptomatic mitral valve stenosis. Once symptoms develop related to pulmonary hypertension 10-year survival is 0–15%. Progressive heart failure is the most common cause death in the untreated patients with mitral stenosis.
Treatment
Medical treatment
No medical treatments will relief mitral stenosis. Dyspnea may be transiently relieved by diuretics or long acting nitrates. In order to slow the heart rate, Beta-blockers or calcium-channel blockers can be prescribed. Patients with atrial fibrillation should be on anticoagulants with a target INR of 2-3. In patients with severe mitral stenosis, cardioversion should not be performed prior to intervention since it does not durably restore sinus rhythm. After successful intervention cardioversion is indicated if atrial fibrillation is of recent onset and the left atrium only moderately enlarged.
Percutaneous mitral commissurotomy
Mitral stenosis can be relieved by percutaneous mitral commisurotomy. This procedure was introduced in 1980 and used worldwide ever since VahanianChen. The results of balloon valvotomy are comparable to open commissurotomy.Reyes
The commissural splitting of percutaneous mitral commissurotomy substantially increases the valvular area. Pulmonary pressures decrease immediately. Procedural mortality has been reported 0–3%. In 2-10% leaflet tearing results in severe mitral regurgitation. Preprocedural condition of the patients and the experience of the operator and operating team are of major influence on the complication rate of percutaneous mitral commissurotomy. Longterm results of a successful procedure have been reported with event-free survival ranges from 35–70% after 10–15 years.
Surgical intervention
Open commissurotomy or valve replacement is indicated if balloon valvotomy is unfeasible.
The concept of surgical repair of the mitral valve was first introduced in 1902 by Sir Thomas Lauder Brunton, a Scottish physician.( Brunton L, Edin MD: Preliminary note on the possibility of treating mitral stenosis by surgical methods. Lancet 1902; 1:352Chen1). The first successful mitral valve operation was a transventricular commissurotomy by Elliot Cutler in 1923. The following transventricular valvultomies performed with the cardiovalvulotome resulted in regurgitation and Cutler abandoned the procedure.
In 1948, Dwight Harken reported his series of patients with mitral stenosis successfully treated with a valvuloplasty procedure.Harken
After development of cardiopulmonary bypass, the closed commissurotomy has been replaced by an open mitral commissurotomy. This allows not only correction of commissural fusion, but also chordal and papillary fusion.
Surgical intervention can improve the functional capacity and long-term survival of patients with mitral stenosis substantially. Survival rates of 96% and freedom from valve-related complications of 92% at 15 years have been reported.Antunes Surgery should be performed before New York Heart Association (NYHA) class III symptoms are present.
Mitral regurgitation
Mitral valve regurgitation results from inadequate mitral leaflet coaptation during systole. This allows the systolic regurgitation of blood from the high-pressure LV to the normally low-pressure LA. The regurgitating volume depends on both the size of the regurgitant orifice and the pressure gradient between the left ventricle and the left atrium. In primary mitral regurgitation, inadequate mitral leaflet coaptation results from an abnormality in any of the functional components of the mitral apparatus. Secondary or functional mitral regurgitation results from left ventricle disease and remodeling.
During systole, combined papillary muscle contraction and contraction of the dynamic annulus promote leaflet coaptation. Calcification of the annulus may hinder the sphincter-like contraction of the annulus allowing regurgitation. Secondary mitral regurgitation due to annulus dilation may be caused by ischemic or dilated cardiomyopathy. The regurgitant volume causes left ventricular enlargement and contractile dysfunction. Left ventricle dilation may cause enlargement of the mitral annulus and the regurgitant orifice, increasing the mitral regurgitation. Positive inotropes, diuretics and vasodilators reduce the size of the left ventricle and the regurgitant orifice, and decrease the regurgitant flow.
Etiology |
Three different types of primary mitral regurgitation can be defined; leaflet retraction from fibrosis and calcification, annular dilatation and chordal abnormalities (including rupture, elongation, or shortening). Functional mitral regurgitation results from LV dysfunction with or without annular dilation. Mitral regurgitation was classified by Carpentier into three types based on leaflet and chordal motion: normal leaflet motion (type I), leaflet prolapse or excessive motion (type II), and restricted leaflet motion (type III).FannIngelsMiller
Chronic mitral valve regurgitation |
Degenerative mitral valve disease is the most common cause of mitral regurgitation in Europe.
Myxomatous mitral valve degeneration is also known as floppy mitral valve or mitral valve prolapse. Prolapse is defined as excursion of one or both leaflets above the plane of the annulus during systole.Gilbert Prolapse of the middle portion of the posterior leaflet is the most common finding in degenerative MR.Carpentier
Mitral regurgitation in Barlow syndrome or parachute mitral valve is due to annular dilatation and extensive hooding of leaflets with large amounts of excessive leaflet tissue. In mitral regurgitation, the mass-to-volume ratio of the enlarged, thin walled left ventricle is less than one.FeiringRumberger
Clinical Presentation |
Patients with mild to moderate compensated chronic mitral regurgitation may remain asymptomatic for many years. The adapted left ventricle maintains normal forward cardiac output. The left ventricle ejection fraction in chronic mitral regurgitation may be greater than normal due to the compensatory cardiac adaptations. Progression of severity depends on etiology of regurgitation; in patients with connective tissue disease regurgitation tends to progress more rapidly than patients with mitral valve prolapse or rheumatic mitral regurgitation. Progression in acute rheumatic fever is often rapid. Acute progression may by caused by endocarditis or chordae rupture.
Gradual progression and eventually decompensation results in decreased cardiac output with physical activity and pulmonary congestion. Patients present with weakness, fatigue, palpitations, dyspnea on exertion. Hepatomegaly, peripheral edema and ascites due to right sided heart failure can be associated with rapid clinical deterioration.
Acute mitral regurgitation is associated with sudden pulmonary congestion and edema.
On physical examination the apical impulse is displaced laterally, indicating left ventricular enlargement. S1 is normal or diminished. S2 may be single, closely split, normally split, or even widely split as a consequence of the reduced resistance to LV ejection. A widely split S2 is often audible, due to shortening of LV systole and early closure of the aortic valve. The P2 component of the second heart sound may be increased if pulmonary hypertension has developed. The apical systolic murmur is typically holosystolic and radiates to the axilla, depending on the direction of the regurgitant jet. It can be blowing, moderately harsh, or even soft. An S3 gallop often is present, reflecting the transmitral diastolic flow during the rapid filling phase.
Early in the disease process of patients with Barlow syndrome, a characteristic midsystolic click can be appreciated, followed by a late systolic murmur; with disease progression the murmur becomes holosystolic, and the midsystolic click may become inaudible.FannIngelsMiller
Acute mitral regurgitation |
Immediate intervention is often necessary in acute mitral regurgitation. Etiology can be organic or functional. Organic causes include rupture of a major chorda tendinea (in myxomatous mitral valve disease) or papillary muscle (due to myocardial infarction), leaflet perforation (of endocarditic origin), and dysfunction of a prosthetic valve due to endocarditis or paravalvular regurgitation. Regurgitation of functional etiology results from left ventricular abnormalities such as dyskinetic wall due to ischemia or dilated ventricle due to cardiomyopathy.
Acute mitral regurgitation is associated with dyspnea and orthopnea, caused by sudden pulmonary congestion and edema. Acute papillary muscle rupture may mimic the presentation of a patient with a postinfarction ventricular septal defect.Harrison
On physical examination no signs of cardiac compensatory mechanisms are present. The increase in left atrial pressure diminishes the pressure gradient between the left ventricle and left atrium by midsystole. The murmur of mitral regurgitation is shortened and of decreased intensity, it may be soft, short of even inaudible. An S3 gallop usually is present. The volume overload is increasing the severity of MR over time, and leads to a greater percentage of the LV stroke volume being ejected in a retrograde fashion.
Diagnostic Options |
Chest Radiography
In chronic mitral regurgitation chest radiography demonstrates enlargement of the left ventricle and atrium. The mitral annulus may be calcified. In acute mitral regurgitation, no adaptive left atrium or ventricle enlargement has developed. Signs of interstitial or alveolar pulmonary edema may be present.
Electrocardiography
Signs of left ventricular hypertrophy and left atrial enlargement due to chronic volume overload may be seen on the electrocardiogram. Atrial fibrillation is a common finding late in the natural history of the disease. Q-waves may be seen in ischemic mitral regurgitation. In patients with acute mitral regurgitation, left atrial and ventricular enlargement may not be evident and the electrocardiogram may be normal or show only nonspecific findings. The resting electrocardiogram of patients with asymptomatic mitral valve prolaps is normal. A variety of ST-T-wave changes, including T-wave inversion and sometimes ST-segment depression, particularly in the inferior leads, can be found in patients with symptomatic mitral valve prolapse.
Echocardiography
Mitral valve pathology and pathophysiology is primary assessed by echocardiography. Left atrial and ventricular dimensions can be quantified. The etiology and mechanism of mitral regurgitation can be identified by echocardiography. Leaflet abnormalities and chordal morphology and function is assessed by echocardiography. Fused subvalvular apparatus due to rheumatic valvulitis and leaflet destruction due to endocarditis can be visualized by echocardiography. Myxoid degeneration of the mitral valve is characterized by an excess of tissue and by leaflets of more than 5mm thickness. Annular dilatation is characterized by a ratio of >1.3 anterior–posterior diameter of the annulus to the length of the anterior leaflet in diastole. The regurgitation caused by annular dilatation and incomplete leaflet coaptation is directed straight back into the left atrium.
In ischaemic MR, the apical displacement of the leaflets can be quantified by measuring the tenting area and the leaflet opening angles.
Color doppler analysis can be used to grade the severity of the regurgitation and permits visualization of the origin, extent, direction, duration, and velocity of disturbed backward flow of the regurgitant leak or leaks into the left atrium.FannIngelsMiller MR is considered severe when the jet area is >10cm2 or >40% of the left atrial area.
Cardiac Catheterization
Coronary ischemic causes of mitral regurgitation can be identified by cardiac catheterization.
Treatment |
Surgical
The hemodynamic overload on the heart caused by mitral regurgitation can ultimately only be corrected by surgically restoring valve competence. For all valve surgery timing of surgery is essential. Irreversible left ventricular dysfunction will result in suboptimal results indelayed surgery. Due to the operative risk and risk of valve prosthesis surgery should however be delayed as long as possible
Mitral valve regurgitation is surgically corrected by mitral valve replacement or repair. Mitral valve repair is generally found to be superior to replacement, with preservation of left ventricular function and part of the mitral valve apparatus EnriquezSarano Gillinov Grossi Zalaquett and without the use of a prosthesis. In mitral valve regurgitation indication for valve surgery is influenced by symptomatic status, ventricular functional status, and the procedure to be performed. Repair might be considered in asymptomatic patients with normal left ventricular function or patients with severe impairment of left ventricular function who might not be candidates for mitral valve replacement.
For most patients, mitral valve surgery is performed for the relief of symptoms or to prevent worsening of asymptomatic left ventricular dysfunction.
Tricuspid stenosis
Tricuspid stenosis (TS) is most commonly of rheumatic origin and combined with tricuspid regurgitation. The anatomical characteristics are similar to those of mitral stenosis, including fibrous leaflet thickening and fusion and shortening of the subvalvular apparatus. The preponderance of cases is in young women. Other aetiologies of right atrial obstruction are rare and include congenital tricuspid atresia, right atrial tumors and carcinoid syndrome
The normal valve area of the tricuspid valve is 7–8cm2. Reduction of valve area to <2 cm2 causes a pressure gradient. A small diastolic pressure gradient (<5 mmHg), gradient between the right atrium and ventricle can be present due to tricuspid stenosis. The gradient is increasing on inspiration. A mean pressure gradient >5mmHg is considered indicative of significant TS and is usually associated with symptoms
Clinical presentation
A tricuspid opening snap and a characteristic mid-diastolic murmur may be audible along the left sternoid border on auscultation. Carvallo’s sign, an increase of murmur intensity on inspiration, may be present. Distention of jugular veins, ascites, pleural effusion and peripheral edema may be present due to increased right atrial pressures.
Reduced cardiac output causes symptoms of fatigue and malaise. The pulmonary congestion of mitral stenosis may be masked in severe tricuspid stenosis.
Diagnostic Options |
Chest radiography
Cardiomegaly with an increase in right atria and pulmonary artery size is demonstrated on chest radiography.
Electrocardiography
An increased P-wave amplitude is seen on the electrocardiogram if the patient is in normal sinus rhythm.
Echocardiography
The tricuspid valve structure and function is commonly assessed by echocardiography. The annular size can be measured and the right pressures can be evaluated.
Tricuspid stenosis due to rheumatic disease is characterized by leaflet thickening with reduced motion and frequent commissural fusion. The chordae are shortened and thickened, and diastolic doming is present. Carcinoid syndrome is characterized by retraction of leaflets towards the apex during systole. A prolonged slope of antegrade flow across the tricuspid valve can be seen on Doppler. Tricuspid stenosis is considered severe when the mean transvalvular gradient is >5 mmHg.
Treatment |
The therapeutic approach for tricuspid regurgitation is dictated by the aetiology of the regurgitation and overall condition of the patient. In a limited number of patients percutaneous balloon tricuspid dilatation has been performed. This is a treatment option in cases of isolated and pure tricuspid stenosis, but it frequently induces regurgitation.Vahanian1 Tricuspid balloon valvotomy, combining commissurotomy leaflet augmentation and annuloplasty, can be used to treat tricuspid stenosis; however, with this treatment the potential for inducing severe tricuspid regurgitation still exists. A biological prosthesis is preferred in case of tricuspid valve replacement,since it heas satisfactory long-term durability and mechanical prosthesis caries a higher risk of thrombosis.
Tricuspid regurgitation
Functional tricuspid regurgitation results from distortion of the architecture and coordinated actions of the tricuspid leaflets, annulus, chords, papillary muscles, and right ventricular (RV) wall. This distortion is most commonly caused by right ventricular dilation and dysfunction from left sided heart disease with pressure/volume overload conditions.
Pressure volume overload conditions cause enlargement of the tricuspid annulus and the shaddle shape becomes more circular. The normal annular excursion can be reduced by 50% in severe tricuspid regurgitation.Fukuda Functional tricuspid regurgitation is a marker of poor prognosis in patients with left ventricular cardiomyopathy. Pure tricuspid regurgitation may result from rheumatic fever, infective endocarditis, carcinoid causes, rheumatoid arthritis, radiation therapy, anorectic drugs, trauma, Marfan’s syndrome, tricuspid valve prolapse, papillary muscle dysfunction, or congenital disorders.
Right ventricular infarction with severe regional wall motion abnormality or disruption of the papillary muscle may cause regurgitation. Right ventricular dilatation with annular enlargement and valvular incompetence can be seen in Eisenmenger syndrome and pulmonary hypertension.
Clinical presentation |
A reduction in cardiac output related to tricuspid regurgitation, may cause symptoms of fatigue and weakness. Right-sided heart failure may cause ascites, congestive hepatosplenomegaly, pulsatile liver, pleural effusions, and peripheral edema.
With progression of the disease, patients become cachexic, cyanotic and jaundice may be present. A parasternal pansytolic murmur increasing on inspiration may be appreciated on auscultation(Carvallo’s sign). An S3, increasing with inspiration and decreasing with a Valsalva maneuve may be audible, as well as an increased P2 if pulmonary hypertension has developed.
Diagnostic options |
Chest Radiography
Cardiomegaly, increased right atrial and ventricular size and a prominent azygous vein can be demonstrated on chest x-ray. Chest Radiography may reveal pleural effusion, and ascites by upward diaphragmatic displacement.
Echocardiography
The tricuspid valve structure and function can be assessed echocardiographically and specific abnormalities can be identified. Distinction between primary and functional forms of tricuspid regurgitation can be made with echocardiography. The annular size can be measured. This imaging modality is used to evaluate the degree of regurgitation, pressures and ventricular function. Severe regurgitation is characterized by systolic flow reversal in the hepatic veins and a vena contracta diameter of more than 7 mm.
Cardiac catheterization
Cardiac catheterization is not necessary to diagnose tricuspid regurgitation. Increased right atrial and right ventricular end-diastolic pressures and the degree of pulmonary artery hypertension can be evaluated by catheterization. Pulmonary artery pressures of over 60 mmHg are usually due to left-sided lesions leading to secondary tricuspid regurgitation.
Treatment |
Treatment strategy for tricuspid regurgitation is dictated by the aetiology and the underlying cause of the disease and the overall condition of the patient. Primary tricuspid regurgitation has a poor prognosis without treatment. Functional tricuspid regurgitation may improve following treatment of its cause. Correction of concomitant mitral regurgitation may worsen tricuspid regurgitation. Risk factors for persistence or worsening of tricuspid regurgitation are reduced right ventricular function and the diameter of tricuspid annulus.
Medical
When pulmonary hypertension is the underlying cause of tricuspid annular dilation, medical management alone may minimize the need for surgical intervention
Surgical
Surgical treatment can be recommended for primary tricuspid regurgitation, when there is pulmonary hypertension, important dilatation of the annulus (diameter >40mm or >21mm/m2).
Surgical options for tricuspid regurgitation include annuloplasty or valve replacement with a mechanical valve or bioprosthesis. Functional tricuspid regurgitation may be repaired by suture annuloplasty (De Vega procedure) or by ring annuloplasty. Longterm outcomes of ring annuloplasty are superior to those of suture annuloplasty. Annuloplasty can be optimized by the use of intraoperative transesophageal echocardiography. Bioprostheses are generally preferred above mechanical prostheses for the tricuspid position, as mentioned in the section about tricuspid stenosis.
Pulmonary valve stenosis
Etiology and pathology |
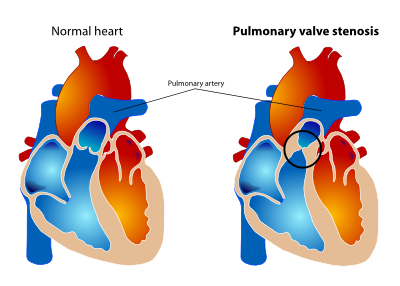
Pulmonary valve stenosis can be caused congenital, carcinoid and rheumatic disorders or extrinsic compression. The typical domeshaped pulmonary valve stenosis is the most common form of right ventricular outflow tract obstruction. Stenosis is caused by fusion of the pulmonary valve leaflets and a narrowed central orifice. The valve is usually mobile and associated with medial abnormalities and dilation of the pulmonary trunk. Pulmonary valve stenosis may be associated with Noonan, Williams, Alagille, Keutel or rubella syndromes.Elizabeth
Clinical presentation |
Most patients with mild to moderate pulmonary valve stenosis are asymptomatic. Severe pulmonary valve stenosis may cause exertional dyspnea and fatigue, chest pain, palpitations and syncope. On physical examination, thrill along the left sternal edge, and a long systolic ejection murmur with late peak may be appreciated. S2 may be widely split with reduced or absent P2.
Diagnostic options |
Chest Radiography
Chest radiography may show dilated pulmonary arteries, occasionally with calcification of the pulmonic valve. In case of severe pulmonary valve stenoisis, oligemic lung fields can be seen.
Electrocardiography
Right ventricular hypertrophy can be seen on electrocardiography as an axis deviation to the right. Right bundle branch block may also be present. Patients with Noonan syndrome invariably have a left bundle branch block.
Echocardiography
Transthoracic echocardiography confirms the diagnosis. The level of obstruction, valvular, supravalvular or right ventricular outflow tract, can be identified. Valvular stenosis is characterized by mildly thickened leaflets with restricted systolic excursion. The pressure gradient can be measured. Associated cardiac conditions can be demonstrated with echocardiography.
Cardiac catheterization
Cardiac catheterization is not necessary to diagnose pulmonic valve stenosis. Hemodynamic consequences and severity of pulmonary valve stenosis can be assessed with cardiac catheterization.
Treatment |
Invasive intervention is recommended in case of symptomatic disease, or when the gradient across the valve is >40 mmHg.
Medical
Supportive and symptomatic treatment of right ventricular failure is recommended.
Surgical
The treatment of choice for stenosis at the valvular level is balloon valvuloplasty. Long-term results are satisfactory and the procedure relatively safe. Surgical valvotomy is very effective with minimal recurrence, however significant pulmonary regurgitation may occur. Pulmonary valve replacement is indicated if the patient is not suitable for balloon valvuloplasty or surgical valvotomy.
Pulmonary valve regurgitation
Etiology and pathology |
Physiologic pulmonic regurgitation, qualified as trace to mild, is present in nearly all individuals. Pulmonary valve regurgitation can be caused by valvular disease such as infective endocarditis (rarely involves pulmonic valve) or connective tissue disease, carcinoid, congenital heart disease, or it can be secondary to pulmonary hypertension, which causes dilation of the valve ring. Pulmonic regurgitation can result in impairment of right ventricular function and eventual clinical manifestations of right-sided volume overload and heart failure.
Clinical presentation |
Patients are often asymptomatic. Symptoms of right-sided heart failure develop when the severity and duration of the regurgitation results in right ventricular enlargement and decompensation. Symptoms include dyspnea on exertion, light-headedness, lethargy, peripheral edema, chest pain, palpitations, and abdominal pain. Often these symptoms are accepted by the patient and attributed to poor physical fitness, causing a delay in presentation. The jugular venous pressure is usually increased in pulmonary valve regurgitation. A palpable impulse may be present at the left lower sternal border due to right ventricular enlargement. On auscultation P2 may be delayed due to increased right ventricular end-diastolic volume and increased ejection time with large stroke volume. P2 can be accentuated in case of pulmonary hypertension, The murmur of pulmonary regurgitation is heard best at the third to fourth intercostal space along the left sternal border and increases with inspiration.
Diagnostic options |
Chest Radiography
Chest radiography of patients with pulmonic regurgitation with tricuspid regurgitation may demonstrate cardiomegaly and enlargement of the right-sided heart contour. Pure pulmonic regurgitation may to have specific signs on chest radiography. Prominent central pulmonary arteries with enlarged hilar vessels and loss of vascularity in the peripheral lung fields suggest severe pulmonary hypertension.
Electrocardiography
Signs of right ventricular hypertrophy can be seen on the Electrocardiogram if pulmonary hypertension is present including a tall R wave in V1 or qR in V1, R wave greater than S wave in V1, R wave progression reversal in the precordial leads and Right axis deviation. A right bundle branch block can be present.
Echocardiography
Echodoppler is the main diagnostic tool for recognizing pulmonic regurgitation. Regurgitant jet and velocity is visualized by Doppler. The width of the regurgitating jet can be used to quantify the severity. Diastolic regurgitation as well as early peak flow velocity in systole suggests the presence of pulmonary hypertension. Echocardiography can reveal right ventricular hypertrophy and dilatation. Right ventricular volume overload is characterized by abnormal septal wall motion. Structural abnormalities of the pulmonic valve or congenital absence of the valve can be demonstrated by echocardiography.
Treatment |
The right ventricle normally adapts to low-pressure volume, high-pressure volume overload in contrast, ultimately leads to heart failure. Determining the underlying cause of pulmonic regurgitation and possible coexisting pulmonary hypertension is essential for appropriate therapy. Treating the cause of pulmonary hypertension can relieve symptoms and decrease the severity of PR If medical management is insufficient, surgical treatment options should be evaluated. The presence of severe right heart failure due to pulmonic regurgitation surgical pulmonic valve reconstruction or replacement can be considered.
References
<biblio>
- Bonow pmid=9870202
- Bonow2 pmid=18820172
- Bonow3 pmid=18848134
- Brener pmid=7829781
- BurgeDeHoratius pmid=8416130
- CarapetisCurrieMathews pmid=10813149
- Carpentier pmid=6887954
- Chaikof pmid=17914037
- DeHumaniCorporisFabrica
- Di pmid=22192280
- Emery Emery R Wi et al. "Aortic Valve Replacement With a Mechanical Cardiac Valve Prosthesis." Cardiac Surgery in the Adult. Ed. Cohn L.H. Third ed. New York: McGraw-Hill, 2012. 841-56.
- EnriquezSarano pmid=15745978
- FannIngelsMiller Fann J.I., Ingels N.B.jr, and Miller D.Ci. "Pathophysiology of Mitral Valve Disease." Cardiac Surgery in the Adult. Ed. Cohn LH. third ed. New York: McGraw-Hill, 2012. 973-1012.
- Fedak pmid=12186790
- FeiringRumberger pmid=1532542
- Fukuda pmid=16820625
- Gilbert pmid=975465
- Gillinov pmid=18374775
- Grossi pmid=11726886
- Iung pmid=17976510
- Michelena pmid=18506017
- Mihaljevic Mihaljevic T. et al. "Pathophysiology of Aortic Valve Disease." Cardiac Surgery in the Adult. Ed. Cohn L.H. Third Edition ed. McGraw-Hill Education, 2012. 825-40.
- MillWilcoxAnderson Mill M.R., Wilcox B.R., and Anderson R.H. "Surgical Anatomy of the Heart." Cardiac Surgery in the Adult. Ed. Cohn LH. New York: McGraw-Hill, 2012. 29-50.
- Mohler pmid=11257079
- Muresian pmid=18773480
- Nishimura pmid=18702976
- Nkomo pmid=16980116
- OBrien pmid=12390952
- Otto pmid=10403851
- Otto2 pmid=18815402
- OttoBurwaskLegget Otto CM, Burwash IG, Legget ME et al. Prospective study of asymptomatic valvular aortic stenosis. Clinical, echocardiographic, and exercise predictors of outcome. Circulation 1997 May 6;95(9):2262-70.
- PaderaSchoen Padera R Fi J r and Schoen F Ji. "Pathology of Cardiac Surgery." Cardiac Surgery in the Adult. Ed. Cohn L.H. New York: McGraw-Hill, 2012. 111-78.
- Rajamannan pmid=22007101
- Rajamannan2 pmid=18703687
- RajamannanGershBonow pmid=12807865
- SiuSilversides pmid=20579534
- SolerGalve pmid=10814642
- Stephenson Stephenson L Wi. "History of Cardiac Surgery." Cardiac Surgery in the Adult. Ed. Cohn L.H. Third Edition ed. New York: McGraw-Hill, 2012. 3-28.
- Stewart pmid=9060903
- Stritzke pmid=19608594
- Thanassoulis pmid=18941438
- Tzemos pmid=18799444
- Vitale pmid=15224997
- Warnes pmid=19038677
- Zalaquett pmid=16341364
- VahanianIung Alec Vahanian, Bernard Iung, Luc Piérard, Robert Dion, John Pepper. Valvular Heart Disease . In: A.JOHN CAMM, THOMAS F.LUSCHER, PATRICK W.SERRUYS, editors. The ESC Textbook of Cardiovascular Medicine. third ed. Oxford University Press; 2012.
- Vahanian Vahanian A, Cormier B, Iung B. Mitral valvuloplasty. In: Topol, editor. Textbook of Interventional Cardiology. 5th ed. Philadelphia: Saunders Elsevier; 2012. p. 879-93.
- Chen Chen CR, Cheng TO. Percutaneous balloon mitral valvuloplasty by the Inoue technique: a multicenter study of 4832 patients in China. Am Heart J 1995 June;129(6):1197-203.
- Reyes Reyes VP, Raju BS, Wynne J et al. Percutaneous balloon valvuloplasty compared with open surgical commissurotomy for mitral stenosis. N Engl J Med 1994 October 13;331(15):961-7.
- Chen1 Chen F Y, Cohn LH. Mitral Valve Repair. In: Cohn LH, editor. Cardiac surgery in the adult. Third ed. New York: McGraw-Hill; 2012. p. 1013-30.
- Harken HARKEN DE, ELLIS LB, . The surgical treatment of mitral stenosis; valvuloplasty. N Engl J Med 1948 November 25;239(22):801-9.
- Antunes Antunes MJ, Vieira H, Ferrao de OJ. Open mitral commissurotomy: the 'golden standard'. J Heart Valve Dis 2000 July;9(4):472-7.
- Harrison Harrison MR, MacPhail B, Gurley JC et al. Usefulness of color Doppler flow imaging to distinguish ventricular septal defect from acute mitral regurgitation complicating acute myocardial infarction. Am J Cardiol 1989 October 1;64(12):697-701.
- Vahanian1 Vahanian A, Palacios IF. Percutaneous approaches to valvular disease. Circulation 2004 April 6;109(13):1572-9.
- Elizabeth Elisabeth Bédard, Michael A Gatzoulis. Adult congenital heart disease. In: S.Yusuf JACAJCELFaBJG, editor. evidence based cardiology. third ed. 2012.
- IungB Iung B, Baron G, Tornos P, Gohlke-Barwolf C, Butchart EG, Vahanian A. Valvular heart disease in the community: a European experience. Curr Probl Cardiol 2007 November;32(11):609-61.
- Cribier Cribier A, Eltchaninoff H, Bash A et al. Percutaneous transcatheter implantation of an aortic valve prosthesis for calcific aortic stenosis: first human case description. Circulation 2002 December 10;106(24):3006-8.
</biblio>