Myocardial Infarction
Each year, more than 2 million Americans have a heart attack or stroke, and more than 800,000 of them die; cardiovascular disease is the leading cause of death in the United States and the largest cause of lower life expectancy among blacks.
Pathophysiology
An acute coronary syndrome (ACS) is most commonly caused by rupture or erosion of an atherosclerotic plaque with superimposed thrombus formation. The underlying process is atherosclerosis, a chronic disease in which artery walls thicken by deposition of fatty materials such as cholesterol and inflammatory cells. The accumulation of this material results in the formation of an atherosclerotic plaque, encapsulated by connective tissue, which can narrow the lumen of the arteries significantly and progressively causing symptoms as angina pectoris or lead to an ACS. Depending on the presence of myocardial damage and typical ECG characteristics, ACS can be divided into ST-segment elevation myocardial infarction (STEMI), and non-ST-segment ACS including non-ST-segment elevation MI (NSTEMI) and unstable angina. In the case of STEMI and NSTEMI, there is biochemical evidence of myocardial damage (infarction). REFNAME1
History
The most typical characteristic of an ACS is acute prolonged chest pain. REFNAME2 The pain does not decrease at rest and is only temporarily relieved with nitroglycerin. Frequent accompanying symptoms include a radiating pain to shoulder, arm, back and/or jaw. REFNAME3 Shortness of breath can occur, as well as sweating, fainting, nausea and vomiting, so called vegetative symptoms. Some patient groups like elderly and diabetics might present with aspecific symptoms. REFNAME4, REFNAME5
It is important to complete the history with information about the medical history (prior history of ischemic events or vascular disease), risk factors for cardiovascular disease (a.o. diabetes mellitus, current smoking, hypertension, hyperlipidemia) and family history (first degree relatives with myocardial infarction before the age 55 of (males) or 65 (females) and/or sudden cardiac death). REFNAME6
Symptoms of heart failure such as orthopnea (dyspnoea when lying flat), progressive dyspnoea and peripheral oedema are indicative for the extent of the problem. REFNAME7
Physical Examination
The focus of the physical examination should be to recognize signs of systemic hypoperfusion such as hypotension, tachycardia, impaired cognition, pale and ashen skin. REFNAME8
Furthermore, signs of heart failure are important, such as pulmonary crackles during auscultation and pitting oedema of the ankles.
In more stable ACS patients, history and physical examination are helpful to exclude other causes of chest pain, such as angina pectoris, aorta dissection, arrhythmias, pulmonary embolism, pneumonia, heartburn, hyperventilation or musculoskeletal problems. REFNAME9
Electrocardiogram (ECG)
An electrocardiogram (ECG) should be made within 10 minutes of arrival in every patient with suspected ACS. REFNAME10
The ECG is an important and easy modality which can assist in the diagnosis and prognostication of ACS.
It can however take 90 minutes after the onset of the symptoms to see abnormalities on the ECG. Furthermore, the ECG does not reflect the dynamic pathophysiology of the ACS. Therefore it is important to make serial ECGs, certainly if a patient has ongoing symptoms. REFNAME11
Furthermore, the ECG is also helpful in localising the ischemia:
- Anterior wall ischemia - One or more of leads V1-V6
- Anteroseptal ischemia - Leads V1 to V3
- Apical or lateral ischemia - Leads aVL and I, and leads V4 to V6
- Inferior wall ischemia - Leads II, III, and aVF
- Posterior wall – Leads V7-V9
- Right ventricle – Leads V3R, V4R
- Left main coronary artery ischemia – Lead aVR
Cardiac Markers
Cardiac markers are essential in order to confirm the diagnosis of MI, indicated by elevated Creatine Kinase isoenzyme MB (CK MB) and/or (high-sensitive) troponins. Troponins are more specific and sensitive than CK MB. The cardiac troponin concentration begins to rise around 4 hours after the onset of myocardial cell damage. REFNAME12
With high-sensitive troponins, myocardial cell damage can be detected even earlier.
It can take 4-6 hours before the CK MB concentration is elevated. Serial measurements are advised in order to estimate infarct size and increase the sensitivity of the (older) assays. REFNAME13
A pitfall concerning mildly elevated cardiac markers can be patients with renal failure or pulmonary embolism. REFNAME14 Although cardiac markers are helpful for confirming the diagnosis, reperfusion should not always wait till the cardiac markers are known if the ECG or symptoms are evident.
Treatment
As the formation of an intracoronary thrombus is the most common cause of the ACS and (recurrent) subsequent outcomes, the cornerstone in the treatment of ACS is antithrombotic treatment. All patients diagnosed with ACS should start with aspirin and a P2Y12 receptor blocker (clopidogrel, prasugrel or ticagrelor). REFNAME15 Aspirin and the P2Y12 receptor blocker are both platelet aggregation inhibitors. The treatment of ACS also focuses on medication to keep the workload of the heart as low as possible. β blockers lower heart rate and blood pressure, to decrease the oxygen demand of the heart. REFNAME16 Nitrates dilatate the coronary arteries so the heart receives more oxygenated blood. REFNAME17
Depending on the (working) diagnosis STEMI or NSTE-ACS, the reperfusion strategy differs.
ST-Segment Elevation Myocardial Infarction
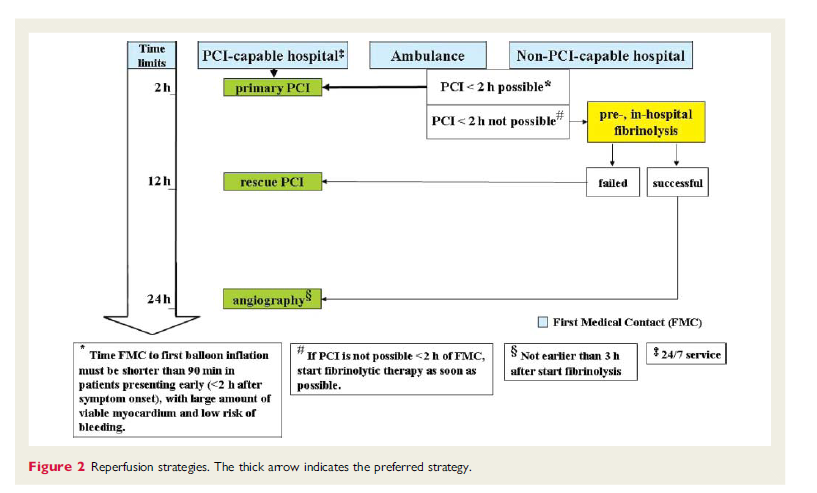
Initial treatment of STEMI is relief of ischemic pain, stabilization of the hemodynamic status, restoration of coronary flow, and myocardial tissue perfusion. Reperfusion therapy should be initiated as quickly as possible within 12 hours after symptom onset by preferably primary percutaneous coronary intervention (PCI) or otherwise fibrinolysis. Meanwhile other measures as continuous cardiac monitoring, oxygen and intravenous access are necessary to guarantee the safety of the patient. REFNAME18
Rapid revascularisation is essential to minimize the size of the myocardial infarction and thereby reduce mortality. In the first hours after symptom onset the amount of salvageable myocardium by reperfusion is greatest. REFNAME19, REFNAME20 There is no consensus whether reperfusion after 12 hours from the onset of symptoms is still beneficial.
Primary PCI is the preferred revascularisation method for patients with STEMI. It is an effective method of securing and maintaining coronary patency and avoids the higher bleeding risk associated with fibrinolysis. If a patient is referred to a non-PCI-capable hospital, and transfer to a PCI-capable hospital in order to perform PCI within 2 hours after the onset of symptoms is not possible, fibrinolytic therapy is recommended.
There are some circumstances in which transfer to a PCI qualified hospital is essential:
- Patients with contraindications for fibrinolysis, such as: active bleedings, recent dental surgery, past history of intracranial bleeding. REFNAME21
- Patients with cardiogenic shock, severe heart failure and/or pulmonary oedema complicating the myocardial infarction. REFNAME22, REFNAME23
Available data support the pre-hospital initiation of fibrinolytics if this reperfusion strategy is indicated. Fibrinolytics like streptokinase stimulate the conversion of plasminogen to plasmin. Plasmin demolishes fibrin which is an important constituent of the thrombus. Fibrinolytics are most effective the first hours after the onset of symptoms, and a benefit is observed in terms of reducing mortality within the first twelve hours. REFNAME24 The hazards of thrombolysis are the increased bleeding risk, including strokes. Because re occlusion after fibrinolysis is possible patients should be transferred to a PCI qualified hospital once fibrinolysis is done. REFNAME25
There are circumstances in which CABG could be indicated, such as failed fibrinolysis and/or PCI, when the patient develops cardiogenic shock, life threatening ventricular arrhythmias, has three vessel disease, or mechanical complications of the MI. REFNAME26
Non-ST-Segment Elevation Acute Coronary Syndrome
Comparable to STEMI, revascularization in NSTE-ACS relieves symptoms, shortens hospital stay, and improves prognosis. However, NSTE-ACS patients represent a heterogenous population, and indication and timing of revascularization depend on many factors, including the baseline risk of the patient. According to current guidelines, depending on early risk stratification a choice has to be made between a routine invasive or a selective invasive (or “conservative strategy”) REFNAME27
Early risk stratification is helpful to identify patients at high risk who might benefit the most from a more aggressive therapeutic approach in order to prevent further ischemic events. REFNAME28
Early risk stratification can be performed using one of the validated risk scores, such as the TIMI risk score:
- - Age ≥65 years
- - Presence of at least three risk factors for coronary heart disease (hypertension, diabetes, dyslipidemia, smoking, or positive family history of early MI)
- - Prior coronary stenosis of ≥50 percent
- - Presence of ST segment deviation on admission ECG
- - At least two anginal episodes in prior 24 hours
- - Elevated serum cardiac biomarkers
- - Use of aspirin in the prior seven days
Patients with a score of 0 to 1 are at low risk, score 2 to 3 are at intermediate risk, score 4 to 6 are at high risk.
Regarding treatment strategies in NSTE-ACS, many randomized controlled trials (RCTs) and meta-analyses have assessed the effects of a routine invasive vs. conservative or selective invasive approach in the short and long term. One of the most recent meta-analysis has shown a benefit of the routine invasive management that was mainly visisble in intermediate- to high-risk patients. (referentie)
Selective Invasive (“or Conservative”) Management
Patients undergoing a selective invasive (“or conservative”) management are initially stabilized by optimal medication, including aspirin and clopidogrel orally and nitro-glycerine, heparin and a beta blocker intravenously. If the patients becomes unstable or has refractory angina, he/she is referred for coronary angiography. Patients stabilized on medical therapy should undergo ischemia detection test before discharge. Potential advantages of this treatment strategy are a reduction of the number of catherization procedures. A potential disadvantage is a prolonged stay in the hospital. Although meta-analyses suggest the superiority of a routine invasive management, trials in which the selective invasive strategy was characterized by high rates of revascularization show equivalence of the two strategies.
Routine Invasive Management
The routine invasive strategy consists of routine, early coronary angiography (tijd noemen, ik dacht < 24 uur) and subsequent revascularization by PCI or CABG based on the angiographic findings.
The optimal timing of coronary angiography with an intended routine invasive management is still a topic for debate.
References
<biblio>
- REFNAME1 PMID 11084798
- REFNAME2 PMID 16304077
- REFNAME3 PMID 10099685
- REFNAME4 PMID 10866870
- REFNAME5 PMID 10751787
- REFNAME6 PMID 15138242
- REFNAME7 PMID 15289388
- REFNAME8 PMID 15289388
- REFNAME9 PMID 15289388
- REFNAME10 PMID 15289388
- REFNAME11 PMID 15289388
- REFNAME12 PMID 16556688
- REFNAME13 PMID 7702648
- REFNAME14 PMID 17951284
- REFNAME15 PMID 21873419
- REFNAME16 PMID 16735367
- REFNAME17 PMID 3925741
- REFNAME18 PMID 15289388
- REFNAME19 PMID 8712096
- REFNAME20 PMID 16311237
- REFNAME21 PMID 14532318
- REFNAME22 PMID 16186438
- REFNAME23 PMID 12472924
- REFNAME24 PMID 16311237
- REFNAME25 PMID 15769784
- REFNAME26 PMID 18191746
- REFNAME27 PMID 15289388
- REFNAME28 PMID 10938172
</biblio>