Bradycardia
Sébastien Krul, MD
Introduction
| Abbreviations |
|
|
|
|
|
|
Bradycardias are symptomatic heart rhythm disorders resulting from an inappropriately low heart rhythm due to inappropriate slow impulse formation or conduction delay of the cardiac impulse in the myocardium or conduction system during physiologic conditions. These two problems can lead to a slow heart rate, a bradycardia. Generally the definition of bradycardia is a heart rate of <60 beats per minute. However, a normal variation of heart rate existsSpodick. For instance, during sleep and in athletes the heart rate can be as low as 40 beats per minute.Talan
Bradycardia can be caused by a variety of intrinsic and extrinsic causes. The most common intrinsic cause is ageing, but ischemic heart disease, infiltrative diseases or surgery can also result in conduction disorders.Ferrer, Mangrum, ESC Medication that modifies the excitability of the heart is the most frequent extrinsic cause. However, electrolyte and metabolic disorders may influence the heart rate directly or indirect.
Complaints from bradycardia result from an insufficient capacity of the heart to supply the body with blood. Complaints of palpitations, syncope or heart failure may result from bradyarrhythmias, but often vague symptoms like dizziness, exercise intolerance or fatigue may be more prominentMova. A causal relation between complaints and the bradycardia should be established and reversible causes should be identified (for instance use of certain drugs).
Disorders of Conduction and Impulse Formation
Sinus Node Dysfunction
Sinus Bradycardia
Sinus bradycardia is a slow sinus rhythm of <60 beats per minuteSpodick. Sinus bradycardia can be physiological, as in athletes or during sleepEctor2. Commonly sinus bradycardia is caused by medication, ischemia or neuro-mediated bradycardia, such as in a vasovagal reactionESC. Furthermore metabolic diseases can cause bradycardia, e.g. hypothermia or hypothyroidism.
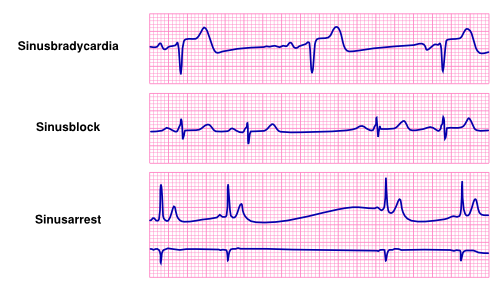
Sinus Node Exit Block
In the case of sinus node exit block, an impulse generated from the sinus node is blocked at one of the exit sides of the sinus node. However impulse formation is not affected, therefore the interval between subsequent beats should be similar to n=x times the P-P interval. On the surface electrocardiogram this is expressed as a pause.
Like AV-nodal conduction disorders (see below) multiple subtypes can be distinguished:
- Second degree Type I (Wenkebach) SA exit block: the P-P interval progressively shortens prior to the pause
- Second degree Type II SA exit block: the pause equals approximately 2-4 times the preceding PP interval
- Third degree SA exit block: absence of P waves, but still impulse formation at the level of the sinus node (can only be diagnosed with an sinus node electrode, during electrophysiological evaluation)
Sinus Arrest
If the sinus node has a problem with impulse formation it is defined as a sinus arrest. There can be the appearance of an irregular rhythm, however sinus P-waves are clearly present, between intervals of no rhythm or an escape rhythm. In comparison with the sinus node exit block, there is no relation with a previous P-P interval. Often an ectopic pacemaker takes over lower in the conduction system, but the new rate varies slightly from the old one.
Asystole
Asystole is the lack of cardiac activity eventually leading to immediate death.
Sick Sinus Syndrome
Sick sinus syndrome is a denoter of diseases of inappropriate sinus node responses Ferrer. These encompass for instance:
- An inappropriate response after tachycardia due to overdrive suppression (which can result in long pauses)
- An inadequate response to exercise.
- Bradycardia-tachycardia syndrome; where alternating bradycardia and tachycardia arise.
AV-Block
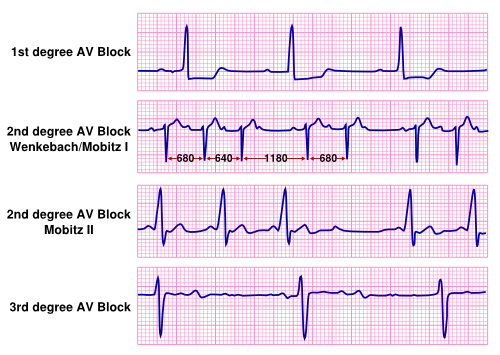
First Degree AV Block
Technically an AV-delay and not an AV block, 1st degree AV block is defined as a prolonged interval between atrial and ventricular activation (>200ms). This delay results from disease in the AV-node or His-Purkinje system. An AV block is not the cause of bradycardia, because every atrial impulse results in conduction to the ventricles.
Second Degree AV Block
The second degree AV block can be divided in two separate entities depending on the clinical characteristics of the conduction disorder. If conduction to the ventricle is conducted in a 2:1 fashion; that is if after every second P-wave there is no conduction to the ventricle, it is not possible to distinguish between the two types and a severe kind of conduction block should be assumed. If two sequential P-wave are not followed by a QRS-complex the term malignant block is used, as this could lead to or be an indication of a total block.
- Mobitz I (Wenkebach): The Mobitz type I block is characterized by a progressively increased P-Q interval until atrial activation is blocked in the AV-node. Thereafter conduction is restored and this cycle repeats itself. A common finding in the Mobitz I block is that the first prolongation of the PR interval is associated with the largest increase in interval. After this first prolongation of the interval, the interval gradually increases. Usually Mobitz type II block is located at the atrioventricular node and rarely deteriorates to a more severe conduction block.Simons, Strasberg
- Mobitz II: When atrial activation is blocked, without progressively increasing P-Q interval a Mobitz Type II AV block is present. This sudden failure of AV conduction is an omen of severe conduction disease in usually infra-Hision part of the atrioventricular conduction system.Simons, Zipes, Donoso
Third Degree AV Block
Third degree AV block is complete block of conduction between atria en ventricle. Atrial and ventricular rhythms are complete dissociated.Levine
Paroxysmal AV block
Paroxysmal atrioventricular block (PAVB), is characterized by a sudden and unexpected block of the atrial impulse. Due to the delayed emerge of an escape rhythm, these patients often present with syncope. However, if a escape rhythm is established patients may present themselves without symptoms. Two different variations of the PAVB are commonly distinguished;
Ventricular Conduction Block
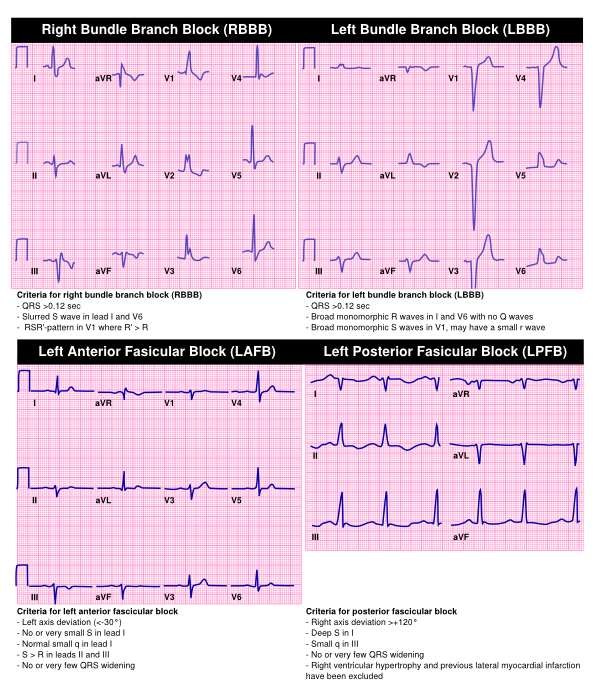
Right bundel branch block (RBBB)
The right bundle branch is composed of one fascicle. Right bundle branch block is a unifascicular block of the right bundle and can be found in healthy people and is represented by a broad QRS complex (>120ms). However a new RBBB in a patient with a history of normal ventricular conduction warrens further cardiological investigation. The last activity is to the right and results in a RSR’ pattern in V1 where R’ > R. This results from the delayed activation of the right ventricle. In V6 a slurred S wave can be seen at the end of the QRS complex.
Left anterior fascicular block (LAFB)
The left bundle branch is composed of two fascicles. One of the fascicle has an anterior location and activates the interventricular septum and the anterior of the ventricle. Clinically this is represented by a left axis deviation and an absent or very small S and normal q in lead I and a S>R in lead II and III. QRS duration should be <120ms.
Left posterior fascicular block (LPFB)
The second fascicle of the left bundle branch is the posterior fascicle. This fascicle has a posterior location and activates the posterior and lateral part of the ventricle. This fascicular block results in a right axis deviation and is represented by a deep S in I and small q in III with a QRS duration of <120ms.
Left bundel branch block (LBBB)
If the two fascicles of the left bundle branch show conduction block there is a left bundle branch block, this bifascicular block is uncommon in healthy patients and further cardiologic investigations need to be performed to screen for underlying disease. Left bundle branch block causes the left ventricle to activate later then the right ventricle. This results in typical ECG characteristics, most importantly a broad QRS of >120ms. In V1 a broad monomorphic S wave can be seen (sometimes with a small r wave) representing slow left ventricular activation. In the lead V6 a broad monomorphic R wave is seen with no Q waves.
Functional conduction disorders: Aberrant Conduction
In certain conditions a conduction disorder can arise on the ECG. This functional conduction disorder is called aberrant conduction and can mimic any form of interventricular conduction disorder. In most cases a right bundle branch block pattern can be seen on the surface ECG because the right bundle has a longer refractory period. There are a few mechanisms which can cause aberrant conduction.
Phase 3 aberration
Phase 3 aberration is a situation that occurs when the bundle branches receive a new impulse, before they are repolarized. The bundles are still in their refractory period. This is also called Ashman phenomenon. As a result of the refractionary state of the bundle, conduction can not proceed along the refractory bundle and a conduction block is visible on the surface ECG. Thus for instance short coupled atrial activity can procedure phase 3 abberant conduction.
Phase 4 abberation or deceleration dependant
During a prolonged interval the Purkinje fibers can depolarize spontaneously. This depolarisation results in the conduction slowing and can even produce a conduction block. This is usually a pathological response, resulting from an increased activity in the Purkinje fibers, but can be normal at very low heart rates. Phase 4 aberration thus only occurs after prolonged pause.
Acceleration dependant
This kind of aberrant conduction resembles phase 3 aberrant conduction; however appearance of the conduction disorders is the result of a small increase in rhythm. The conduction disorder is a result of an abnormal response of tissue that has diminished excitability.
Diagnosis
In the diagnosis of bradyarrhythmias the identification of reversible causes is important to prevent unnecessary treatment. After a detailed history and physical examination there are additional investigations which can give information about the location of the nature of the bradyarrhythmia. While not all investigations are necessary, a thorough work-up has to be performed to prevent serious clinical events or pacemaker implantation. Espaccially the differentiation between bradyarrhtyhmias and vasovagal syncope can be difficult, but is very important for the management of symptoms.
Symptoms
A patient with a bradyarrhythmia can be complete asymptomatic. However patients with bradycardia may present with a diversity of signs and symptoms. A complete cardiac standstill can result in syncope (pause >6 sec) or near syncope. More often symptoms are nonspecific and chronic and are a result of the chronotopic incompetence and reduced cardiac output. Symptoms like for example, dizziness, light-headedness or confusional states, episodes of fatigue or muscular weakness, exercise intolerance, heart failure or palpitations can be described by the patient.
Investigations
There are a number of additional investigations which can uncover the cause of bradyarrhythmias.
- ECG: A surface ECG can demonstrate the conduction disorder and relate complaints to electrocardiographic findings. A Valsalva manoeuvre or carotid sinus massage whilst performing an ECG can give information about function of the autonomous nervous system and its possible role in the occurrence of the bradyarrrhythmia.
- X-ECG: An exercise test can give information about the chronotropic competence of the cardiac conduction system.
- Long-term ECG recording: Holter recording can identify causes of paroxysmal or intermittent bradyarrhythmias. Importantly a correlation with symptoms can be made and pathological causes of bradyarrhythmias or long pause (>3sec) during the night can be identified. If 24h or 48h Holter recordings cannot identify the cause of symptoms longer duration of monitoring may be required. Transient event recorders can record up to 30seconds of ECG when a patient activates the device. This device can be especially useful when non-invasive monitoring is required due to the low occurrence of the bradyarrhythmia. For longer monitoring an implantable loop recorder can be used. This small device can be implanted and observe rhythm over an extensive period.
- Electrophysiological testing: If non-invasive testing does not discover the bradyarrhythmia underlying the symptoms and electrophysiologic study may be undertaken to assess sinus nodal function and atrioventricular conduction. The measurement of conduction intervals and reaction to standard electrophysiological pacing protocols can elucidate the cause of bradyarrhtyhmia.
Treatment
Fortunately the human heart has a couple of backup mechanisms that can sustain a heart rate in case of severe bradycardia. These escape mechanisms can occur in every part of the heart (i.e. atrium, AV, node, ventricle). In general, the rate of the escape mechanism is faster when the escape rhythm is located higher in the conduction system. If no reversible cause for the bradyarrhythmia can be found and the bradyarrhythmia persist, further therapy is required if the patients remains symptomatic.
Drug Therapy
There are no options for chronic drug therapy in bradyarrhthmias. In the acute setting atropine or isoprenaline may be used to increase heart rate or AV-nodal conduction. Patients with severe bradyarrhythmias (Type 2 AV nodal Mobitz II block, Type 3 AV nodal block, sinus arrest >3 seconds) should be considered for permanent or temporary pacing therapy.
Temporary pacing
Temporary pacing can be used to bridge the time to pacemaker implantation or until the bradyarrhythmia is resolved. Transvenous pacing is the most accepted method and can be used to pace the right atrium or the right ventricle after insertion of a temporary pacemaker wire through venous access. Pacing through the oesophagus can only capture the atrium, due to the anatomical position of the heart in relation with the oesophagus. Transcutanous pacing is a painful and emergency option in which muscle and heart are stimulated with large electrodes. Finally epicardal pacing is usually performed after cardiac surgery and requires surgical implantation of the electrodes.
Device Therapy
Implantable pacemakers activate cardiac myocardium with electrical stimulation, leading to muscle contraction. Due to the nature of a pacemaker, the activation is different from the physiological conduction system, there are electrical and mechanical consequences. It is therefore important to adjust pacemaker setting to the individual patient. The type of pacemakers and their settings are extensively covered in the device chapter of cardiac arrhythmias. The indications for pacemaker implantation in patients with bradyarrhythmias are mentioned below.
- Sinus node disease: Pacemaker implantation should be considered in patients with sinus node disease which manifests as symptomatic bradycardia in which the symptom-rhythm correlation must have been 1) spontaneously occurring or 2) drug-induced where alternative drug therapy is lacking. Furthermore other eligible candidates for permanent pacing are patients with syncope with sinus node disease, either spontaneously occurring or induced at electrophysiological study and sinus node disease manifests as symptomatic chronotropic incompetence which is 1) spontaneously occurring or 2) drug-induced where alternative drug therapy. Patients with sinus node disease without symptoms including use of bradycardia-provoking drugs, ECG findings of sinus node dysfunction with symptoms not due directly or indirectly to bradycardia or symptomatic sinus node dysfunction where symptoms can reliably be attributed to non-essential medication do not have an indication for permanent pacemaker therapy.
- Atrioventricular Block: The following patients with AV conduction block have an indication for pacemaker therapy; 1) chronic symptomatic third or second degree (Mobitz I or II) atrioventricular block 2) neuromuscular diseases (e.g. myotonic muscular dystrophy etc.) with third-degree or second-degree atrioventricular Block or 3) third or second degree (Mobitz I or II) atrioventricular block after catheter ablation of the atrioventricular junction or after valve surgery when the block is not expected to resolve. Patients with asymptomatic first degree atrioventricular block, asymptomatic second degree Mobitz I with supra-Hisian conduction block or atrioventricular block expected to resolve do not require a pacemaker implantation.
- Intraventricular conduction Block: Patient which show a intermittent third-degree atrioventricular block, second-degree Mobitz II atrioventricular block. alternating bundle branch block or findings on electrophysiological study of markedly prolonged His-Ventrical interval (≥100ms) or pacing-induced infra-His block in patients with symptoms have an indication for pacemaker therapy. Patients with a bundle branch block without atrioventricular block or symptoms and bundle branch block with first-degree atrioventricular block without symptoms should not have a pacemaker implanted.
References
<biblio>
- Epstein pmid=23255456
- Ferrer pmid=5695590
- Talan pmid=7083929
- Mangrum pmid=10706901
- Strasberg pmid 7471363
- Donoso pmid 14118480
- Zipes pmid 378457
- Levine pmid=13356435
- Kay pmid=6461235
- Kastor pmid=1089890
- Dreifus pmid=6826942
- Ector pmid=6191291
- Langberg pmid 2598419
- Kim pmid=11230857
- Glikson pmid=9388104
- Shaw pmid=4005079
- Choksk pmid=2360528
- Mymin pmid=3762641
- Stevenson pmid=2299071
- James pmid=14451031
- Friedberg pmid=14206803
- Dhingra pmid=4817704
- McAnulty1 pmid=7088050
- McAnulty2 pmid=619828
- Spodick pmid=1529897
- Ector2 pmid=6147639
- Mova pmid=19713422
- Simons pmid=9894656
- Wiens pmid=6741841
- Lichstein pmid=6176956
- Kinley pmid=7503472
- Krahn pmid=15125724
- Hoffman pmid=3536438
- ESC isbn=9780199566990
- ECGpedia http://en.ecgpedia.org
- Robles isbn=9789031313983
- Wellens isbn=9781416002598
- Elsherrif PMCID: PMC2877697